| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Prostaglandin E2 receptor EP3 subtype |
|---|
| Ligand | BDBM50303695 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_596846 (CHEMBL1042657) |
|---|
| IC50 | 2±n/a nM |
|---|
| Citation |  Singh, J; Zeller, W; Zhou, N; Hategan, G; Mishra, RK; Polozov, A; Yu, P; Onua, E; Zhang, J; Ramírez, JL; Sigthorsson, G; Thorsteinnsdottir, M; Kiselyov, AS; Zembower, DE; Andrésson, T; Gurney, ME Structure-activity relationship studies leading to the identification of (2E)-3-[l-[(2,4-dichlorophenyl)methyl]-5-fluoro-3-methyl-lH-indol-7-yl]-N-[(4,5-dichloro-2-thienyl)sulfonyl]-2-propenamide (DG-041), a potent and selective prostanoid EP3 receptor antagonist, as a novel antiplatelet agent that J Med Chem53:18-36 (2010) [PubMed] Article Singh, J; Zeller, W; Zhou, N; Hategan, G; Mishra, RK; Polozov, A; Yu, P; Onua, E; Zhang, J; Ramírez, JL; Sigthorsson, G; Thorsteinnsdottir, M; Kiselyov, AS; Zembower, DE; Andrésson, T; Gurney, ME Structure-activity relationship studies leading to the identification of (2E)-3-[l-[(2,4-dichlorophenyl)methyl]-5-fluoro-3-methyl-lH-indol-7-yl]-N-[(4,5-dichloro-2-thienyl)sulfonyl]-2-propenamide (DG-041), a potent and selective prostanoid EP3 receptor antagonist, as a novel antiplatelet agent that J Med Chem53:18-36 (2010) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Prostaglandin E2 receptor EP3 subtype |
|---|
| Name: | Prostaglandin E2 receptor EP3 subtype |
|---|
| Synonyms: | PE2R3_HUMAN | PGE receptor, EP3 subtype | PGE2-R | PTGER3 | Prostaglandin E2 receptor | Prostaglandin E2 receptor EP3 subtype | Prostaglandin E2 receptor EP3 subtype (EP3) | Prostaglandin E2 receptor EP3A subtype (EP3A) | Prostaglandin E2 receptor EP3D subtype (EP3D) | Prostanoid EP3 receptor |
|---|
| Type: | Enzyme |
|---|
| Mol. Mass.: | 43335.03 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | P43115 |
|---|
| Residue: | 390 |
|---|
| Sequence: | MKETRGYGGDAPFCTRLNHSYTGMWAPERSAEARGNLTRPPGSGEDCGSVSVAFPITMLL
TGFVGNALAMLLVSRSYRRRESKRKKSFLLCIGWLALTDLVGQLLTTPVVIVVYLSKQRW
EHIDPSGRLCTFFGLTMTVFGLSSLFIASAMAVERALAIRAPHWYASHMKTRATRAVLLG
VWLAVLAFALLPVLGVGQYTVQWPGTWCFISTGRGGNGTSSSHNWGNLFFASAFAFLGLL
ALTVTFSCNLATIKALVSRCRAKATASQSSAQWGRITTETAIQLMGIMCVLSVCWSPLLI
MMLKMIFNQTSVEHCKTHTEKQKECNFFLIAVRLASLNQILDPWVYLLLRKILLRKFCQI
RYHTNNYASSSTSLPCQCSSTLMWSDHLER
|
|
|
|---|
| BDBM50303695 |
|---|
| n/a |
|---|
| Name | BDBM50303695 |
|---|
| Synonyms: | CHEMBL585581 | Thiophene-2-sulfonic Acid{(E)-3-[1-(2,4-Dichlorobenzyl)-1Hindol-7-y l]acryloyl}amide |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C22H16Cl2N2O3S2 |
|---|
| Mol. Mass. | 491.41 |
|---|
| SMILES | Clc1ccc(Cn2ccc3cccc(\C=C\C(=O)NS(=O)(=O)c4cccs4)c23)c(Cl)c1 |
|---|
| Structure | 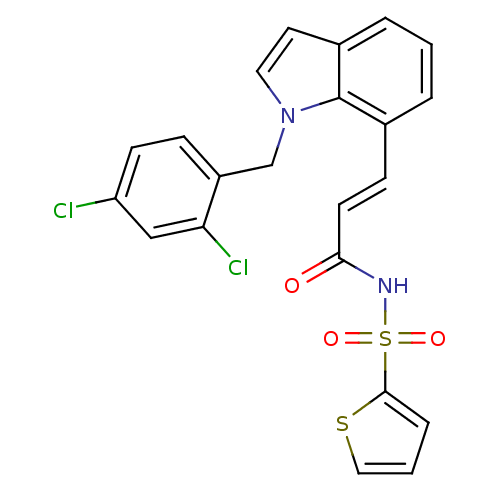
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Singh, J; Zeller, W; Zhou, N; Hategan, G; Mishra, RK; Polozov, A; Yu, P; Onua, E; Zhang, J; Ramírez, JL; Sigthorsson, G; Thorsteinnsdottir, M; Kiselyov, AS; Zembower, DE; Andrésson, T; Gurney, ME Structure-activity relationship studies leading to the identification of (2E)-3-[l-[(2,4-dichlorophenyl)methyl]-5-fluoro-3-methyl-lH-indol-7-yl]-N-[(4,5-dichloro-2-thienyl)sulfonyl]-2-propenamide (DG-041), a potent and selective prostanoid EP3 receptor antagonist, as a novel antiplatelet agent that J Med Chem53:18-36 (2010) [PubMed] Article
Singh, J; Zeller, W; Zhou, N; Hategan, G; Mishra, RK; Polozov, A; Yu, P; Onua, E; Zhang, J; Ramírez, JL; Sigthorsson, G; Thorsteinnsdottir, M; Kiselyov, AS; Zembower, DE; Andrésson, T; Gurney, ME Structure-activity relationship studies leading to the identification of (2E)-3-[l-[(2,4-dichlorophenyl)methyl]-5-fluoro-3-methyl-lH-indol-7-yl]-N-[(4,5-dichloro-2-thienyl)sulfonyl]-2-propenamide (DG-041), a potent and selective prostanoid EP3 receptor antagonist, as a novel antiplatelet agent that J Med Chem53:18-36 (2010) [PubMed] Article