| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Neuroendocrine convertase 2 |
|---|
| Ligand | BDBM50303776 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_597568 (CHEMBL1049705) |
|---|
| Ki | 55±n/a nM |
|---|
| Citation |  Becker, GL; Sielaff, F; Than, ME; Lindberg, I; Routhier, S; Day, R; Lu, Y; Garten, W; Steinmetzer, T Potent inhibitors of furin and furin-like proprotein convertases containing decarboxylated P1 arginine mimetics. J Med Chem53:1067-75 (2010) [PubMed] Article Becker, GL; Sielaff, F; Than, ME; Lindberg, I; Routhier, S; Day, R; Lu, Y; Garten, W; Steinmetzer, T Potent inhibitors of furin and furin-like proprotein convertases containing decarboxylated P1 arginine mimetics. J Med Chem53:1067-75 (2010) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Neuroendocrine convertase 2 |
|---|
| Name: | Neuroendocrine convertase 2 |
|---|
| Synonyms: | KEX2-like endoprotease 2 | NEC 2 | NEC2 | NEC2_HUMAN | Neuroendocrine convertase 2 | PC2 | PCSK2 | Prohormone convertase 2 | Proprotein convertase 2 |
|---|
| Type: | PROTEIN |
|---|
| Mol. Mass.: | 70563.93 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | ChEMBL_1290738 |
|---|
| Residue: | 638 |
|---|
| Sequence: | MKGGCVSQWKAAAGFLFCVMVFASAERPVFTNHFLVELHKGGEDKARQVAAEHGFGVRKL
PFAEGLYHFYHNGLAKAKRRRSLHHKQQLERDPRVKMALQQEGFDRKKRGYRDINEIDIN
MNDPLFTKQWYLINTGQADGTPGLDLNVAEAWELGYTGKGVTIGIMDDGIDYLHPDLASN
YNAEASYDFSSNDPYPYPRYTDDWFNSHGTRCAGEVSAAANNNICGVGVAYNSKVAGIRM
LDQPFMTDIIEASSISHMPQLIDIYSASWGPTDNGKTVDGPRELTLQAMADGVNKGRGGK
GSIYVWASGDGGSYDDCNCDGYASSMWTISINSAINDGRTALYDESCSSTLASTFSNGRK
RNPEAGVATTDLYGNCTLRHSGTSAAAPEAAGVFALALEANLGLTWRDMQHLTVLTSKRN
QLHDEVHQWRRNGVGLEFNHLFGYGVLDAGAMVKMAKDWKTVPERFHCVGGSVQDPEKIP
STGKLVLTLTTDACEGKENFVRYLEHVQAVITVNATRRGDLNINMTSPMGTKSILLSRRP
RDDDSKVGFDKWPFMTTHTWGEDARGTWTLELGFVGSAPQKGVLKEWTLMLHGTQSAPYI
DQVVRDYQSKLAMSKKEELEEELDEAVERSLKSILNKN
|
|
|
|---|
| BDBM50303776 |
|---|
| n/a |
|---|
| Name | BDBM50303776 |
|---|
| Synonyms: | CHEMBL568525 | N-((6S,9S,12S)-1,17-diamino-6-(4-carbamimidoylbenzylcarbamoyl)-1,17-diimino-9-isopropyl-8,11-dioxo-2,7,10,16-tetraazaheptadecan-12-yl)decanamide |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C35H62N12O4 |
|---|
| Mol. Mass. | 714.9448 |
|---|
| SMILES | [#6]-[#6]-[#6]-[#6]-[#6]-[#6]-[#6]-[#6]-[#6]-[#6](=O)-[#7]-[#6@@H](-[#6]-[#6]-[#6]\[#7]=[#6](/[#7])-[#7])-[#6](=O)-[#7]-[#6@@H](-[#6](-[#6])-[#6])-[#6](=O)-[#7]-[#6@@H](-[#6]-[#6]-[#6]\[#7]=[#6](/[#7])-[#7])-[#6](=O)-[#7]-[#6]-c1ccc(cc1)-[#6](-[#7])=[#7] |r| |
|---|
| Structure | 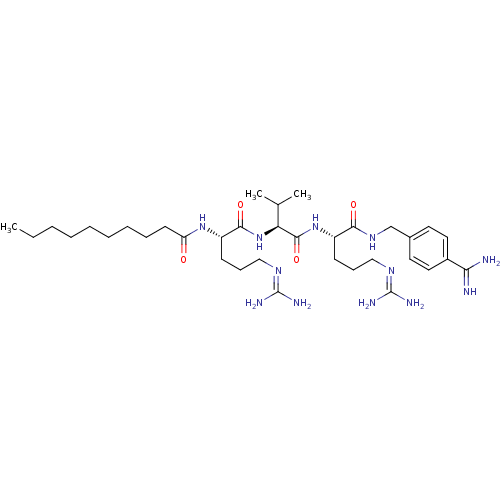
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Becker, GL; Sielaff, F; Than, ME; Lindberg, I; Routhier, S; Day, R; Lu, Y; Garten, W; Steinmetzer, T Potent inhibitors of furin and furin-like proprotein convertases containing decarboxylated P1 arginine mimetics. J Med Chem53:1067-75 (2010) [PubMed] Article
Becker, GL; Sielaff, F; Than, ME; Lindberg, I; Routhier, S; Day, R; Lu, Y; Garten, W; Steinmetzer, T Potent inhibitors of furin and furin-like proprotein convertases containing decarboxylated P1 arginine mimetics. J Med Chem53:1067-75 (2010) [PubMed] Article