

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
| Reaction Details | |||
|---|---|---|---|
 | Report a problem with these data | ||
| Target | Cannabinoid receptor 1 | ||
| Ligand | BDBM50305424 | ||
| Substrate/Competitor | n/a | ||
| Meas. Tech. | ChEMBL_603744 (CHEMBL1039369) | ||
| IC50 | 1.6±n/a nM | ||
| Citation |  Cooper, M; Receveur, JM; Bjurling, E; Nørregaard, PK; Nielsen, PA; Sköld, N; Högberg, T Exploring SAR features in diverse library of 4-cyanomethyl-pyrazole-3-carboxamides suitable for further elaborations as CB1 antagonists. Bioorg Med Chem Lett20:26-30 (2010) [PubMed] Article Cooper, M; Receveur, JM; Bjurling, E; Nørregaard, PK; Nielsen, PA; Sköld, N; Högberg, T Exploring SAR features in diverse library of 4-cyanomethyl-pyrazole-3-carboxamides suitable for further elaborations as CB1 antagonists. Bioorg Med Chem Lett20:26-30 (2010) [PubMed] Article | ||
| More Info.: | Get all data from this article, Assay Method | ||
| Cannabinoid receptor 1 | |||
| Name: | Cannabinoid receptor 1 | ||
| Synonyms: | CANN6 | CANNABINOID CB1 | CB-R | CB1 | CNR | CNR1 | CNR1_HUMAN | Cannabinoid CB1 receptor | Cannabinoid receptor | Cannabinoid receptor 1 (CB1) | Cannabinoid receptor 1 (brain) | ||
| Type: | G Protein-Coupled Receptor (GPCR) | ||
| Mol. Mass.: | 52868.96 | ||
| Organism: | Homo sapiens (Human) | ||
| Description: | P21554 | ||
| Residue: | 472 | ||
| Sequence: |
| ||
| BDBM50305424 | |||
| n/a | |||
| Name | BDBM50305424 | ||
| Synonyms: | CHEMBL599964 | trans-1-(2-chlorophenyl)-5-(4-chlorophenyl)-4-(cyanomethyl)-N-(4-methylcyclohexyl)-1H-pyrazole-3-carboxamide | ||
| Type | Small organic molecule | ||
| Emp. Form. | C25H24Cl2N4O | ||
| Mol. Mass. | 467.39 | ||
| SMILES | C[C@H]1CC[C@@H](CC1)NC(=O)c1nn(c(c1CC#N)-c1ccc(Cl)cc1)-c1ccccc1Cl |r,wU:4.7,wD:1.0,(2.91,-14.12,;2.14,-12.79,;2.9,-11.45,;2.13,-10.11,;.59,-10.13,;-.17,-11.46,;.6,-12.79,;-.19,-8.8,;-1.72,-8.81,;-2.5,-7.48,;-2.49,-10.15,;-1.95,-11.59,;-3.17,-12.55,;-4.45,-11.69,;-4.03,-10.21,;-4.82,-8.89,;-6.36,-8.92,;-7.91,-8.95,;-5.78,-12.47,;-7.11,-11.7,;-8.44,-12.47,;-8.44,-14.02,;-9.78,-14.79,;-7.11,-14.79,;-5.77,-14.02,;-3.11,-14.08,;-4.42,-14.9,;-4.36,-16.44,;-2.99,-17.16,;-1.69,-16.33,;-1.75,-14.79,;-.45,-13.97,)| | ||
| Structure | 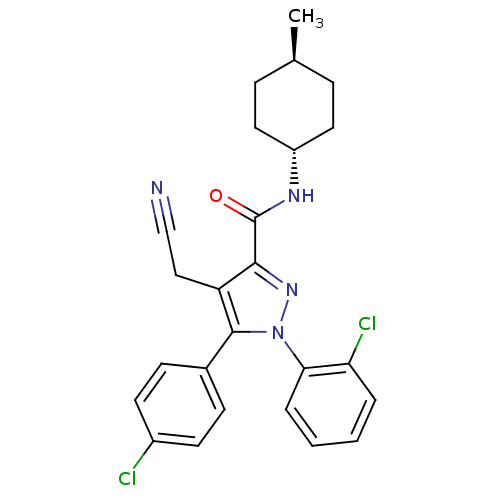
| ||