| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Cytochrome P450 11B2, mitochondrial |
|---|
| Ligand | BDBM7887 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_610340 (CHEMBL1072072) |
|---|
| IC50 | 1799±n/a nM |
|---|
| Citation |  Roumen, L; Peeters, JW; Emmen, JM; Beugels, IP; Custers, EM; de Gooyer, M; Plate, R; Pieterse, K; Hilbers, PA; Smits, JF; Vekemans, JA; Leysen, D; Ottenheijm, HC; Janssen, HM; Hermans, JJ Synthesis, biological evaluation, and molecular modeling of 1-benzyl-1H-imidazoles as selective inhibitors of aldosterone synthase (CYP11B2). J Med Chem53:1712-25 (2010) [PubMed] Article Roumen, L; Peeters, JW; Emmen, JM; Beugels, IP; Custers, EM; de Gooyer, M; Plate, R; Pieterse, K; Hilbers, PA; Smits, JF; Vekemans, JA; Leysen, D; Ottenheijm, HC; Janssen, HM; Hermans, JJ Synthesis, biological evaluation, and molecular modeling of 1-benzyl-1H-imidazoles as selective inhibitors of aldosterone synthase (CYP11B2). J Med Chem53:1712-25 (2010) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Cytochrome P450 11B2, mitochondrial |
|---|
| Name: | Cytochrome P450 11B2, mitochondrial |
|---|
| Synonyms: | Aldosterone Synthase (CYP11B2) | Aldosterone synthase | Aldosterone-synthesizing enzyme | C11B2_HUMAN | CYP11B2 | CYPXIB2 | Cytochrome P450 11B2 | Cytochrome P450 11B2 (CYP11B2) | Cytochrome P450 11B2, mitochondrial | P-450Aldo | P-450C18 | Steroid 18-hydroxylase |
|---|
| Type: | Protein |
|---|
| Mol. Mass.: | 57582.15 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | P19099 |
|---|
| Residue: | 503 |
|---|
| Sequence: | MALRAKAEVCVAAPWLSLQRARALGTRAARAPRTVLPFEAMPQHPGNRWLRLLQIWREQG
YEHLHLEMHQTFQELGPIFRYNLGGPRMVCVMLPEDVEKLQQVDSLHPCRMILEPWVAYR
QHRGHKCGVFLLNGPEWRFNRLRLNPDVLSPKAVQRFLPMVDAVARDFSQALKKKVLQNA
RGSLTLDVQPSIFHYTIEASNLALFGERLGLVGHSPSSASLNFLHALEVMFKSTVQLMFM
PRSLSRWISPKVWKEHFEAWDCIFQYGDNCIQKIYQELAFNRPQHYTGIVAELLLKAELS
LEAIKANSMELTAGSVDTTAFPLLMTLFELARNPDVQQILRQESLAAAASISEHPQKATT
ELPLLRAALKETLRLYPVGLFLERVVSSDLVLQNYHIPAGTLVQVFLYSLGRNAALFPRP
ERYNPQRWLDIRGSGRNFHHVPFGFGMRQCLGRRLAEAEMLLLLHHVLKHFLVETLTQED
IKMVYSFILRPGTSPLLTFRAIN
|
|
|
|---|
| BDBM7887 |
|---|
| n/a |
|---|
| Name | BDBM7887 |
|---|
| Synonyms: | 1-Benzylimidazole (BI) | 1-benzyl-1H-imidazole | 1-benzylimidazole | CHEMBL14192 | US9138393, 1-benzylimidazole |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C10H10N2 |
|---|
| Mol. Mass. | 158.1998 |
|---|
| SMILES | C(c1ccccc1)n1ccnc1 |
|---|
| Structure | 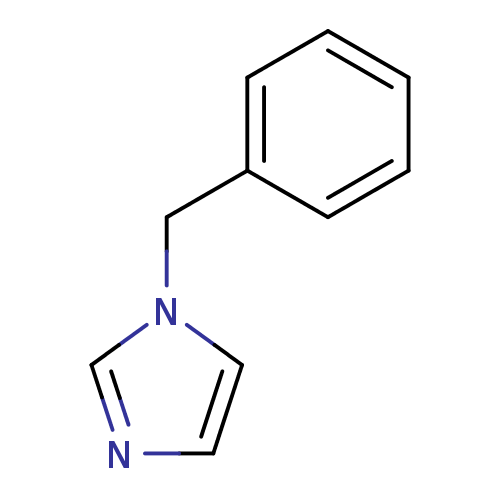
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Roumen, L; Peeters, JW; Emmen, JM; Beugels, IP; Custers, EM; de Gooyer, M; Plate, R; Pieterse, K; Hilbers, PA; Smits, JF; Vekemans, JA; Leysen, D; Ottenheijm, HC; Janssen, HM; Hermans, JJ Synthesis, biological evaluation, and molecular modeling of 1-benzyl-1H-imidazoles as selective inhibitors of aldosterone synthase (CYP11B2). J Med Chem53:1712-25 (2010) [PubMed] Article
Roumen, L; Peeters, JW; Emmen, JM; Beugels, IP; Custers, EM; de Gooyer, M; Plate, R; Pieterse, K; Hilbers, PA; Smits, JF; Vekemans, JA; Leysen, D; Ottenheijm, HC; Janssen, HM; Hermans, JJ Synthesis, biological evaluation, and molecular modeling of 1-benzyl-1H-imidazoles as selective inhibitors of aldosterone synthase (CYP11B2). J Med Chem53:1712-25 (2010) [PubMed] Article