| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Tyrosine-protein kinase Lck |
|---|
| Ligand | BDBM50307506 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_612002 (CHEMBL1072209) |
|---|
| IC50 | >10000±n/a nM |
|---|
| Citation |  Traquandi, G; Ciomei, M; Ballinari, D; Casale, E; Colombo, N; Croci, V; Fiorentini, F; Isacchi, A; Longo, A; Mercurio, C; Panzeri, A; Pastori, W; Pevarello, P; Volpi, D; Roussel, P; Vulpetti, A; Brasca, MG Identification of potent pyrazolo[4,3-h]quinazoline-3-carboxamides as multi-cyclin-dependent kinase inhibitors. J Med Chem53:2171-87 (2010) [PubMed] Article Traquandi, G; Ciomei, M; Ballinari, D; Casale, E; Colombo, N; Croci, V; Fiorentini, F; Isacchi, A; Longo, A; Mercurio, C; Panzeri, A; Pastori, W; Pevarello, P; Volpi, D; Roussel, P; Vulpetti, A; Brasca, MG Identification of potent pyrazolo[4,3-h]quinazoline-3-carboxamides as multi-cyclin-dependent kinase inhibitors. J Med Chem53:2171-87 (2010) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Tyrosine-protein kinase Lck |
|---|
| Name: | Tyrosine-protein kinase Lck |
|---|
| Synonyms: | 2.7.10.2 | LCK | LCK_HUMAN | LSK | Leukocyte C-terminal Src kinase | Lymphocyte cell-specific protein-tyrosine kinase | Lymphocyte-specific protein tyrosine kinase | P56-LCK | Protein YT16 | Proto-oncogene Lck | Proto-oncogene tyrosine-protein kinase LCK | Src/Lck kinase | T cell-specific protein-tyrosine kinase |
|---|
| Type: | n/a |
|---|
| Mol. Mass.: | 57987.83 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | P06239 |
|---|
| Residue: | 509 |
|---|
| Sequence: | MGCGCSSHPEDDWMENIDVCENCHYPIVPLDGKGTLLIRNGSEVRDPLVTYEGSNPPASP
LQDNLVIALHSYEPSHDGDLGFEKGEQLRILEQSGEWWKAQSLTTGQEGFIPFNFVAKAN
SLEPEPWFFKNLSRKDAERQLLAPGNTHGSFLIRESESTAGSFSLSVRDFDQNQGEVVKH
YKIRNLDNGGFYISPRITFPGLHELVRHYTNASDGLCTRLSRPCQTQKPQKPWWEDEWEV
PRETLKLVERLGAGQFGEVWMGYYNGHTKVAVKSLKQGSMSPDAFLAEANLMKQLQHQRL
VRLYAVVTQEPIYIITEYMENGSLVDFLKTPSGIKLTINKLLDMAAQIAEGMAFIEERNY
IHRDLRAANILVSDTLSCKIADFGLARLIEDNEYTAREGAKFPIKWTAPEAINYGTFTIK
SDVWSFGILLTEIVTHGRIPYPGMTNPEVIQNLERGYRMVRPDNCPEELYQLMRLCWKER
PEDRPTFDYLRSVLEDFFTATEGQYQPQP
|
|
|
|---|
| BDBM50307506 |
|---|
| n/a |
|---|
| Name | BDBM50307506 |
|---|
| Synonyms: | 1-Methyl-8-{[1-(phenylcarbonyl)piperidin-4-yl]amino}-4,5-dihydro-1H-pyrazolo[4,3-h]quinazoline-3-carboxamide | CHEMBL599428 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C23H25N7O2 |
|---|
| Mol. Mass. | 431.4903 |
|---|
| SMILES | Cn1nc(C(N)=O)c2CCc3cnc(NC4CCN(CC4)C(=O)c4ccccc4)nc3-c12 |
|---|
| Structure | 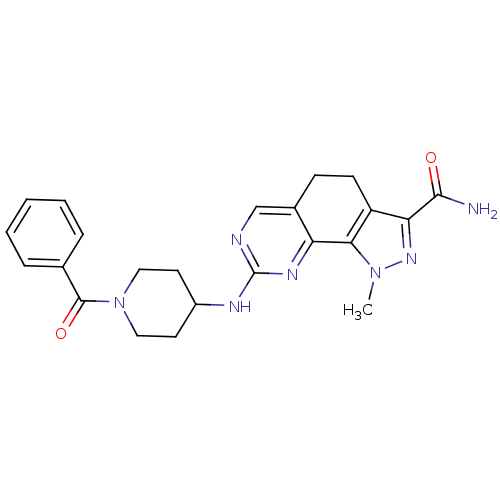
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Traquandi, G; Ciomei, M; Ballinari, D; Casale, E; Colombo, N; Croci, V; Fiorentini, F; Isacchi, A; Longo, A; Mercurio, C; Panzeri, A; Pastori, W; Pevarello, P; Volpi, D; Roussel, P; Vulpetti, A; Brasca, MG Identification of potent pyrazolo[4,3-h]quinazoline-3-carboxamides as multi-cyclin-dependent kinase inhibitors. J Med Chem53:2171-87 (2010) [PubMed] Article
Traquandi, G; Ciomei, M; Ballinari, D; Casale, E; Colombo, N; Croci, V; Fiorentini, F; Isacchi, A; Longo, A; Mercurio, C; Panzeri, A; Pastori, W; Pevarello, P; Volpi, D; Roussel, P; Vulpetti, A; Brasca, MG Identification of potent pyrazolo[4,3-h]quinazoline-3-carboxamides as multi-cyclin-dependent kinase inhibitors. J Med Chem53:2171-87 (2010) [PubMed] Article