| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Histamine H1 receptor |
|---|
| Ligand | BDBM50313618 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_614259 (CHEMBL1106922) |
|---|
| Ki | 1995.26±n/a nM |
|---|
| Citation |  Holmes, IP; Blunt, RJ; Lorthioir, OE; Blowers, SM; Gribble, A; Payne, AH; Stansfield, IG; Wood, M; Woollard, PM; Reavill, C; Howes, CM; Micheli, F; Di Fabio, R; Donati, D; Terreni, S; Hamprecht, D; Arista, L; Worby, A; Watson, SP The identification of a selective dopamine D2 partial agonist, D3 antagonist displaying high levels of brain exposure. Bioorg Med Chem Lett20:2013-6 (2010) [PubMed] Article Holmes, IP; Blunt, RJ; Lorthioir, OE; Blowers, SM; Gribble, A; Payne, AH; Stansfield, IG; Wood, M; Woollard, PM; Reavill, C; Howes, CM; Micheli, F; Di Fabio, R; Donati, D; Terreni, S; Hamprecht, D; Arista, L; Worby, A; Watson, SP The identification of a selective dopamine D2 partial agonist, D3 antagonist displaying high levels of brain exposure. Bioorg Med Chem Lett20:2013-6 (2010) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Histamine H1 receptor |
|---|
| Name: | Histamine H1 receptor |
|---|
| Synonyms: | H1R | HH1R | HISTAMINE H1 | HRH1 | HRH1_HUMAN |
|---|
| Type: | G Protein-Coupled Receptor (GPCR) |
|---|
| Mol. Mass.: | 55808.72 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | Cell pellets from SK-N-MC cells transfected with human H1 receptor were used in binding assay. |
|---|
| Residue: | 487 |
|---|
| Sequence: | MSLPNSSCLLEDKMCEGNKTTMASPQLMPLVVVLSTICLVTVGLNLLVLYAVRSERKLHT
VGNLYIVSLSVADLIVGAVVMPMNILYLLMSKWSLGRPLCLFWLSMDYVASTASIFSVFI
LCIDRYRSVQQPLRYLKYRTKTRASATILGAWFLSFLWVIPILGWNHFMQQTSVRREDKC
ETDFYDVTWFKVMTAIINFYLPTLLMLWFYAKIYKAVRQHCQHRELINRSLPSFSEIKLR
PENPKGDAKKPGKESPWEVLKRKPKDAGGGSVLKSPSQTPKEMKSPVVFSQEDDREVDKL
YCFPLDIVHMQAAAEGSSRDYVAVNRSHGQLKTDEQGLNTHGASEISEDQMLGDSQSFSR
TDSDTTTETAPGKGKLRSGSNTGLDYIKFTWKRLRSHSRQYVSGLHMNRERKAAKQLGFI
MAAFILCWIPYFIFFMVIAFCKNCCNEHLHMFTIWLGYINSTLNPLIYPLCNENFKKTFK
RILHIRS
|
|
|
|---|
| BDBM50313618 |
|---|
| n/a |
|---|
| Name | BDBM50313618 |
|---|
| Synonyms: | 1-(3-chlorophenyl)-3-(piperidin-2-ylmethyl)imidazolidin-2-one | CHEMBL1085672 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C15H20ClN3O |
|---|
| Mol. Mass. | 293.792 |
|---|
| SMILES | Clc1cccc(c1)N1CCN(CC2CCCCN2)C1=O |
|---|
| Structure | 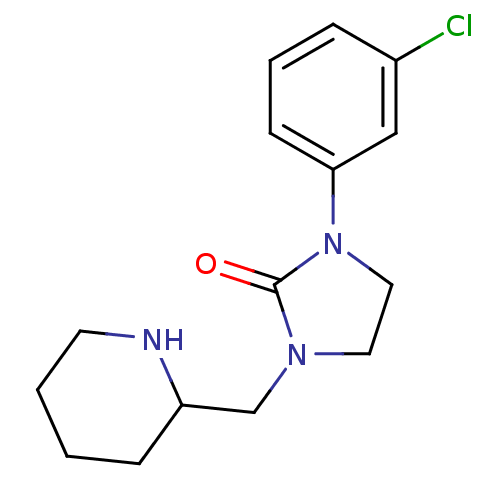
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Holmes, IP; Blunt, RJ; Lorthioir, OE; Blowers, SM; Gribble, A; Payne, AH; Stansfield, IG; Wood, M; Woollard, PM; Reavill, C; Howes, CM; Micheli, F; Di Fabio, R; Donati, D; Terreni, S; Hamprecht, D; Arista, L; Worby, A; Watson, SP The identification of a selective dopamine D2 partial agonist, D3 antagonist displaying high levels of brain exposure. Bioorg Med Chem Lett20:2013-6 (2010) [PubMed] Article
Holmes, IP; Blunt, RJ; Lorthioir, OE; Blowers, SM; Gribble, A; Payne, AH; Stansfield, IG; Wood, M; Woollard, PM; Reavill, C; Howes, CM; Micheli, F; Di Fabio, R; Donati, D; Terreni, S; Hamprecht, D; Arista, L; Worby, A; Watson, SP The identification of a selective dopamine D2 partial agonist, D3 antagonist displaying high levels of brain exposure. Bioorg Med Chem Lett20:2013-6 (2010) [PubMed] Article