| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Corticotropin-releasing factor receptor 1 |
|---|
| Ligand | BDBM50300145 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_615438 (CHEMBL1106905) |
|---|
| IC50 | 0.24±n/a nM |
|---|
| Citation |  Hartz, RA; Ahuja, VT; Schmitz, WD; Molski, TF; Mattson, GK; Lodge, NJ; Bronson, JJ; Macor, JE Synthesis and structure-activity relationships of N3-pyridylpyrazinones as corticotropin-releasing factor-1 (CRF1) receptor antagonists. Bioorg Med Chem Lett20:1890-4 (2010) [PubMed] Article Hartz, RA; Ahuja, VT; Schmitz, WD; Molski, TF; Mattson, GK; Lodge, NJ; Bronson, JJ; Macor, JE Synthesis and structure-activity relationships of N3-pyridylpyrazinones as corticotropin-releasing factor-1 (CRF1) receptor antagonists. Bioorg Med Chem Lett20:1890-4 (2010) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Corticotropin-releasing factor receptor 1 |
|---|
| Name: | Corticotropin-releasing factor receptor 1 |
|---|
| Synonyms: | CRF-R | CRF1 | CRFR1_RAT | CRH-R 1 | Corticotropin releasing factor receptor | Corticotropin releasing factor receptor 1 | Corticotropin-releasing Factor Receptor 1 | Corticotropin-releasing hormone receptor 1 | Crhr | Crhr1 |
|---|
| Type: | G Protein-Coupled Receptor (GPCR) |
|---|
| Mol. Mass.: | 47870.75 |
|---|
| Organism: | Rattus norvegicus (rat) |
|---|
| Description: | Receptor binding assays were performed using rat cortex homogenate. |
|---|
| Residue: | 415 |
|---|
| Sequence: | MGRRPQLRLVKALLLLGLNPVSTSLQDQRCENLSLTSNVSGLQCNASVDLIGTCWPRSPA
GQLVVRPCPAFFYGVRYNTTNNGYRECLANGSWAARVNYSECQEILNEEKKSKVHYHVAV
IINYLGHCISLVALLVAFVLFLRLRSIRCLRNIIHWNLISAFILRNATWFVVQLTVSPEV
HQSNVAWCRLVTAAYNYFHVTNFFWMFGEGCYLHTAIVLTYSTDRLRKWMFVCIGWGVPF
PIIVAWAIGKLHYDNEKCWFGKRPGVYTDYIYQGPMILVLLINFIFLFNIVRILMTKLRA
STTSETIQYRKAVKATLVLLPLLGITYMLFFVNPGEDEVSRVVFIYFNSFLESFQGFFVS
VFYCFLNSEVRSAIRKRWRRWQDKHSIRARVARAMSIPTSPTRVSFHSIKQSTAV
|
|
|
|---|
| BDBM50300145 |
|---|
| n/a |
|---|
| Name | BDBM50300145 |
|---|
| Synonyms: | (S)-5-Chloro-1-(cyclopropyl-2-methoxyethyl)-3-[6-(difluoromethoxy)-2,5-dimethylpyridin-3-ylamino]pyrazin-2(1H)-one | (S)-5-chloro-1-(1-cyclopropyl-2-methoxyethyl)-3-(6-(difluoromethoxy)-2,5-dimethylpyridin-3-ylamino)pyrazin-2(1H)-one | CHEMBL574852 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C18H21ClF2N4O3 |
|---|
| Mol. Mass. | 414.834 |
|---|
| SMILES | COC[C@H](C1CC1)n1cc(Cl)nc(Nc2cc(C)c(OC(F)F)nc2C)c1=O |r| |
|---|
| Structure | 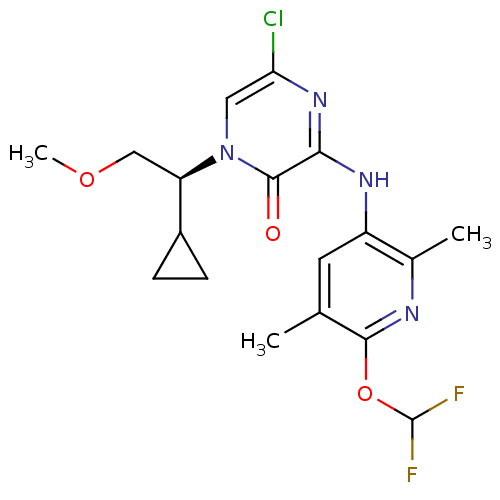
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Hartz, RA; Ahuja, VT; Schmitz, WD; Molski, TF; Mattson, GK; Lodge, NJ; Bronson, JJ; Macor, JE Synthesis and structure-activity relationships of N3-pyridylpyrazinones as corticotropin-releasing factor-1 (CRF1) receptor antagonists. Bioorg Med Chem Lett20:1890-4 (2010) [PubMed] Article
Hartz, RA; Ahuja, VT; Schmitz, WD; Molski, TF; Mattson, GK; Lodge, NJ; Bronson, JJ; Macor, JE Synthesis and structure-activity relationships of N3-pyridylpyrazinones as corticotropin-releasing factor-1 (CRF1) receptor antagonists. Bioorg Med Chem Lett20:1890-4 (2010) [PubMed] Article