| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Beta-secretase 1 |
|---|
| Ligand | BDBM50315011 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_624424 (CHEMBL1108752) |
|---|
| IC50 | 810±n/a nM |
|---|
| Citation |  Malamas, MS; Barnes, K; Hui, Y; Johnson, M; Lovering, F; Condon, J; Fobare, W; Solvibile, W; Turner, J; Hu, Y; Manas, ES; Fan, K; Olland, A; Chopra, R; Bard, J; Pangalos, MN; Reinhart, P; Robichaud, AJ Novel pyrrolyl 2-aminopyridines as potent and selective human beta-secretase (BACE1) inhibitors. Bioorg Med Chem Lett20:2068-73 (2010) [PubMed] Article Malamas, MS; Barnes, K; Hui, Y; Johnson, M; Lovering, F; Condon, J; Fobare, W; Solvibile, W; Turner, J; Hu, Y; Manas, ES; Fan, K; Olland, A; Chopra, R; Bard, J; Pangalos, MN; Reinhart, P; Robichaud, AJ Novel pyrrolyl 2-aminopyridines as potent and selective human beta-secretase (BACE1) inhibitors. Bioorg Med Chem Lett20:2068-73 (2010) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Beta-secretase 1 |
|---|
| Name: | Beta-secretase 1 |
|---|
| Synonyms: | ASP2 | Asp 2 | Aspartyl protease 2 | BACE | BACE1 | BACE1_HUMAN | Beta-secretase (BACE) | Beta-secretase 1 | Beta-secretase 1 (BACE 1) | Beta-secretase 1 (BACE-1) | Beta-secretase 1 (BACE1) | Beta-site APP cleaving enzyme 1 | Beta-site amyloid precursor protein cleaving enzyme 1 | KIAA1149 | Memapsin-2 | Membrane-associated aspartic protease 2 | beta-Secretase (BACE-1) | beta-Secretase (BACE1) |
|---|
| Type: | Protein |
|---|
| Mol. Mass.: | 55755.10 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | P56817 |
|---|
| Residue: | 501 |
|---|
| Sequence: | MAQALPWLLLWMGAGVLPAHGTQHGIRLPLRSGLGGAPLGLRLPRETDEEPEEPGRRGSF
VEMVDNLRGKSGQGYYVEMTVGSPPQTLNILVDTGSSNFAVGAAPHPFLHRYYQRQLSST
YRDLRKGVYVPYTQGKWEGELGTDLVSIPHGPNVTVRANIAAITESDKFFINGSNWEGIL
GLAYAEIARPDDSLEPFFDSLVKQTHVPNLFSLQLCGAGFPLNQSEVLASVGGSMIIGGI
DHSLYTGSLWYTPIRREWYYEVIIVRVEINGQDLKMDCKEYNYDKSIVDSGTTNLRLPKK
VFEAAVKSIKAASSTEKFPDGFWLGEQLVCWQAGTTPWNIFPVISLYLMGEVTNQSFRIT
ILPQQYLRPVEDVATSQDDCYKFAISQSSTGTVMGAVIMEGFYVVFDRARKRIGFAVSAC
HVHDEFRTAAVEGPFVTLDMEDCGYNIPQTDESTLMTIAYVMAAICALFMLPLCLMVCQW
RCLRCLRQQHDDFADDISLLK
|
|
|
|---|
| BDBM50315011 |
|---|
| n/a |
|---|
| Name | BDBM50315011 |
|---|
| Synonyms: | 6-((2-(2-chlorophenyl)-5-(4-(pyridin-2-yloxy)phenyl)-1H-pyrrol-1-yl)methyl)pyridin-2-amine | CHEMBL1091592 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C27H21ClN4O |
|---|
| Mol. Mass. | 452.935 |
|---|
| SMILES | Nc1cccc(Cn2c(ccc2-c2ccccc2Cl)-c2ccc(Oc3ccccn3)cc2)n1 |
|---|
| Structure | 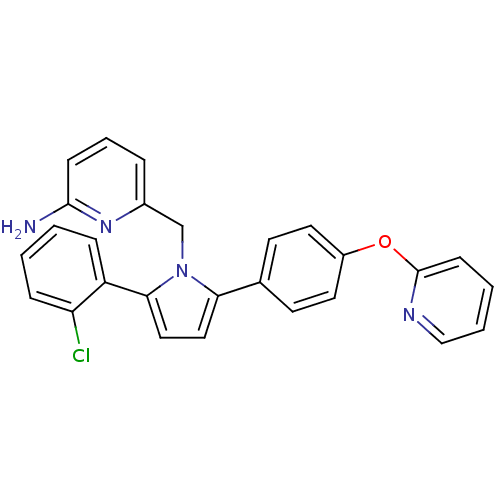
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Malamas, MS; Barnes, K; Hui, Y; Johnson, M; Lovering, F; Condon, J; Fobare, W; Solvibile, W; Turner, J; Hu, Y; Manas, ES; Fan, K; Olland, A; Chopra, R; Bard, J; Pangalos, MN; Reinhart, P; Robichaud, AJ Novel pyrrolyl 2-aminopyridines as potent and selective human beta-secretase (BACE1) inhibitors. Bioorg Med Chem Lett20:2068-73 (2010) [PubMed] Article
Malamas, MS; Barnes, K; Hui, Y; Johnson, M; Lovering, F; Condon, J; Fobare, W; Solvibile, W; Turner, J; Hu, Y; Manas, ES; Fan, K; Olland, A; Chopra, R; Bard, J; Pangalos, MN; Reinhart, P; Robichaud, AJ Novel pyrrolyl 2-aminopyridines as potent and selective human beta-secretase (BACE1) inhibitors. Bioorg Med Chem Lett20:2068-73 (2010) [PubMed] Article