

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
| Reaction Details | |||
|---|---|---|---|
 | Report a problem with these data | ||
| Target | Carbonic anhydrase | ||
| Ligand | BDBM50258714 | ||
| Substrate/Competitor | n/a | ||
| Meas. Tech. | ChEMBL_626038 (CHEMBL1106166) | ||
| Ki | 62±n/a nM | ||
| Citation |  Güzel, O; Maresca, A; Hall, RA; Scozzafava, A; Mastrolorenzo, A; Mühlschlegel, FA; Supuran, CT Carbonic anhydrase inhibitors. The beta-carbonic anhydrases from the fungal pathogens Cryptococcus neoformans and Candida albicans are strongly inhibited by substituted-phenyl-1H-indole-5-sulfonamides. Bioorg Med Chem Lett20:2508-11 (2010) [PubMed] Article Güzel, O; Maresca, A; Hall, RA; Scozzafava, A; Mastrolorenzo, A; Mühlschlegel, FA; Supuran, CT Carbonic anhydrase inhibitors. The beta-carbonic anhydrases from the fungal pathogens Cryptococcus neoformans and Candida albicans are strongly inhibited by substituted-phenyl-1H-indole-5-sulfonamides. Bioorg Med Chem Lett20:2508-11 (2010) [PubMed] Article | ||
| More Info.: | Get all data from this article, Assay Method | ||
| Carbonic anhydrase | |||
| Name: | Carbonic anhydrase | ||
| Synonyms: | CAN_CANAL | NCE103 | beta-Carbonic Anhydrase | ||
| Type: | Enzyme | ||
| Mol. Mass.: | 31587.59 | ||
| Organism: | Candida albicans (Yeast) | ||
| Description: | n/a | ||
| Residue: | 281 | ||
| Sequence: |
| ||
| BDBM50258714 | |||
| n/a | |||
| Name | BDBM50258714 | ||
| Synonyms: | 1-({[5-(Aminosulfonyl)-3-(2-bromophenyl)-1H-indol-2-yl]carbonyl}amino)-2,4,6 trimethylpyridinium perchlorate | 2,4,6-trimethyl-1-(5-sulfamoyl-3-o-tolyl-1H-indole-2-carboxamido)pyridinium perchlorate | CHEMBL466234 | ||
| Type | Small organic molecule | ||
| Emp. Form. | C24H25N4O3S | ||
| Mol. Mass. | 449.545 | ||
| SMILES | Cc1cc(C)[n+](NC(=O)c2[nH]c3ccc(cc3c2-c2ccccc2C)S(N)(=O)=O)c(C)c1 |(7.26,-10.03,;5.72,-10.03,;4.94,-8.69,;3.41,-8.7,;2.63,-7.37,;2.65,-10.03,;1.11,-10.03,;.34,-11.36,;1.11,-12.69,;-1.2,-11.36,;-2.12,-12.62,;-3.59,-12.14,;-4.92,-12.91,;-6.26,-12.14,;-6.26,-10.59,;-4.93,-9.82,;-3.59,-10.59,;-2.12,-10.11,;-1.64,-8.64,;-.13,-8.33,;.34,-6.86,;-.69,-5.72,;-2.2,-6.04,;-2.67,-7.51,;-4.18,-7.83,;-7.59,-9.82,;-8.93,-9.05,;-6.82,-8.49,;-8.35,-11.16,;3.41,-11.36,;2.64,-12.7,;4.95,-11.36,)| | ||
| Structure | 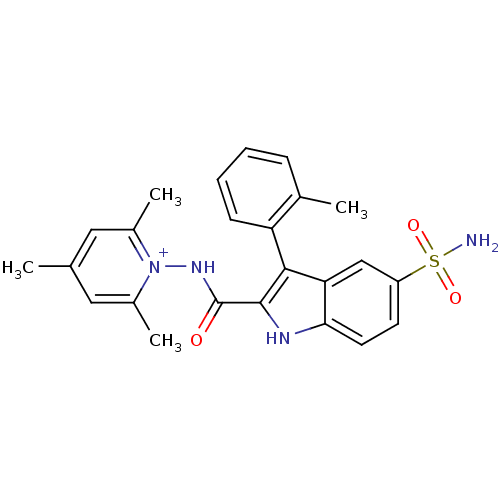
| ||