| Reaction Details |
|---|
 | Report a problem with these data |
| Target | MAP kinase-activated protein kinase 2 |
|---|
| Ligand | BDBM50316253 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_628524 (CHEMBL1104646) |
|---|
| IC50 | >50000±n/a nM |
|---|
| Citation |  Verheijen, JC; Richard, DJ; Curran, K; Kaplan, J; Lefever, M; Nowak, P; Malwitz, DJ; Brooijmans, N; Toral-Barza, L; Zhang, WG; Lucas, J; Hollander, I; Ayral-Kaloustian, S; Mansour, TS; Yu, K; Zask, A Discovery of 4-morpholino-6-aryl-1H-pyrazolo[3,4-d]pyrimidines as highly potent and selective ATP-competitive inhibitors of the mammalian target of rapamycin (mTOR): optimization of the 6-aryl substituent. J Med Chem52:8010-24 (2009) [PubMed] Article Verheijen, JC; Richard, DJ; Curran, K; Kaplan, J; Lefever, M; Nowak, P; Malwitz, DJ; Brooijmans, N; Toral-Barza, L; Zhang, WG; Lucas, J; Hollander, I; Ayral-Kaloustian, S; Mansour, TS; Yu, K; Zask, A Discovery of 4-morpholino-6-aryl-1H-pyrazolo[3,4-d]pyrimidines as highly potent and selective ATP-competitive inhibitors of the mammalian target of rapamycin (mTOR): optimization of the 6-aryl substituent. J Med Chem52:8010-24 (2009) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| MAP kinase-activated protein kinase 2 |
|---|
| Name: | MAP kinase-activated protein kinase 2 |
|---|
| Synonyms: | MAP kinase-activated protein kinase 2 (MAPKAPK2) | MAP kinase-activated protein kinase 2 (MK2) | MAP kinase-activated protein kinase 2 (p38/MK2) | MAPK-Activated Protein Kinase 2 (MK2) | MAPK-activated protein kinase 2 | MAPK2_HUMAN | MAPKAP kinase 2 | MAPKAPK-2 | MAPKAPK2 | MK2 |
|---|
| Type: | Serine/threonine-protein kinase |
|---|
| Mol. Mass.: | 45579.87 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | P49137 |
|---|
| Residue: | 400 |
|---|
| Sequence: | MLSNSQGQSPPVPFPAPAPPPQPPTPALPHPPAQPPPPPPQQFPQFHVKSGLQIKKNAII
DDYKVTSQVLGLGINGKVLQIFNKRTQEKFALKMLQDCPKARREVELHWRASQCPHIVRI
VDVYENLYAGRKCLLIVMECLDGGELFSRIQDRGDQAFTEREASEIMKSIGEAIQYLHSI
NIAHRDVKPENLLYTSKRPNAILKLTDFGFAKETTSHNSLTTPCYTPYYVAPEVLGPEKY
DKSCDMWSLGVIMYILLCGYPPFYSNHGLAISPGMKTRIRMGQYEFPNPEWSEVSEEVKM
LIRNLLKTEPTQRMTITEFMNHPWIMQSTKVPQTPLHTSRVLKEDKERWEDVKEEMTSAL
ATMRVDYEQIKIKKIEDASNPLLLKRRKKARALEAAALAH
|
|
|
|---|
| BDBM50316253 |
|---|
| n/a |
|---|
| Name | BDBM50316253 |
|---|
| Synonyms: | CHEMBL1095627 | tert-Butyl 4-(6-(4-aminophenyl)-4-morpholino-1H-pyrazolo[3,4-d]pyrimidin-1-yl)piperidine-1-carboxylate |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C25H33N7O3 |
|---|
| Mol. Mass. | 479.5746 |
|---|
| SMILES | CC(C)(C)OC(=O)N1CCC(CC1)n1ncc2c(nc(nc12)-c1ccc(N)cc1)N1CCOCC1 |
|---|
| Structure | 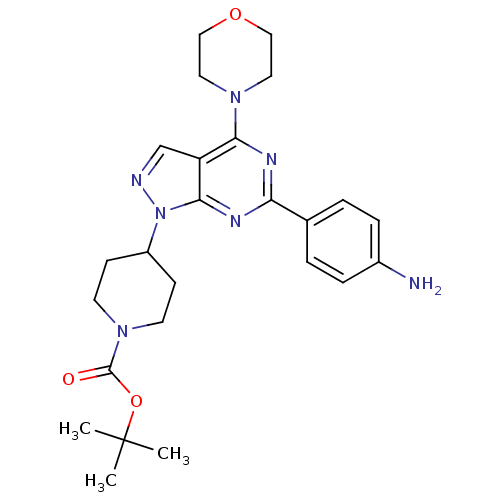
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Verheijen, JC; Richard, DJ; Curran, K; Kaplan, J; Lefever, M; Nowak, P; Malwitz, DJ; Brooijmans, N; Toral-Barza, L; Zhang, WG; Lucas, J; Hollander, I; Ayral-Kaloustian, S; Mansour, TS; Yu, K; Zask, A Discovery of 4-morpholino-6-aryl-1H-pyrazolo[3,4-d]pyrimidines as highly potent and selective ATP-competitive inhibitors of the mammalian target of rapamycin (mTOR): optimization of the 6-aryl substituent. J Med Chem52:8010-24 (2009) [PubMed] Article
Verheijen, JC; Richard, DJ; Curran, K; Kaplan, J; Lefever, M; Nowak, P; Malwitz, DJ; Brooijmans, N; Toral-Barza, L; Zhang, WG; Lucas, J; Hollander, I; Ayral-Kaloustian, S; Mansour, TS; Yu, K; Zask, A Discovery of 4-morpholino-6-aryl-1H-pyrazolo[3,4-d]pyrimidines as highly potent and selective ATP-competitive inhibitors of the mammalian target of rapamycin (mTOR): optimization of the 6-aryl substituent. J Med Chem52:8010-24 (2009) [PubMed] Article