| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Leukotriene B4 receptor 1 |
|---|
| Ligand | BDBM50317625 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_632889 (CHEMBL1107471) |
|---|
| IC50 | 0.21±n/a nM |
|---|
| Citation |  Goodnow, RA; Hicks, A; Sidduri, A; Kowalczyk, A; Dominique, R; Qiao, Q; Lou, JP; Gillespie, P; Fotouhi, N; Tilley, J; Cohen, N; Choudhry, S; Cavallo, G; Tannu, SA; Ventre, JD; Lavelle, D; Tare, NS; Oh, H; Lamb, M; Kurylko, G; Hamid, R; Wright, MB; Pamidimukkala, A; Egan, T; Gubler, U; Hoffman, AF; Wei, X; Li, YL; O'Neil, J; Marcano, R; Pozzani, K; Molinaro, T; Santiago, J; Singer, L; Hargaden, M; Moore, D; Catala, AR; Chao, LC; Hermann, G; Venkat, R; Mancebo, H; Renzetti, LM Discovery of novel and potent leukotriene B4 receptor antagonists. Part 1. J Med Chem53:3502-16 (2010) [PubMed] Article Goodnow, RA; Hicks, A; Sidduri, A; Kowalczyk, A; Dominique, R; Qiao, Q; Lou, JP; Gillespie, P; Fotouhi, N; Tilley, J; Cohen, N; Choudhry, S; Cavallo, G; Tannu, SA; Ventre, JD; Lavelle, D; Tare, NS; Oh, H; Lamb, M; Kurylko, G; Hamid, R; Wright, MB; Pamidimukkala, A; Egan, T; Gubler, U; Hoffman, AF; Wei, X; Li, YL; O'Neil, J; Marcano, R; Pozzani, K; Molinaro, T; Santiago, J; Singer, L; Hargaden, M; Moore, D; Catala, AR; Chao, LC; Hermann, G; Venkat, R; Mancebo, H; Renzetti, LM Discovery of novel and potent leukotriene B4 receptor antagonists. Part 1. J Med Chem53:3502-16 (2010) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Leukotriene B4 receptor 1 |
|---|
| Name: | Leukotriene B4 receptor 1 |
|---|
| Synonyms: | BLT | BLT1 | BLTR | CMKRL1 | Chemoattractant receptor-like 1 | G-protein coupled receptor 16 | GPR16 | LT4R1_HUMAN | LTB4-R 1 | LTB4R | Leukotriene B4 R1 | Leukotriene B4 receptor | Leukotriene B4 receptor 1 | P2RY7 | P2Y purinoceptor 7 | P2Y7 |
|---|
| Type: | Enzyme Catalytic Domain |
|---|
| Mol. Mass.: | 37582.68 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | Q15722 |
|---|
| Residue: | 352 |
|---|
| Sequence: | MNTTSSAAPPSLGVEFISLLAIILLSVALAVGLPGNSFVVWSILKRMQKRSVTALMVLNL
ALADLAVLLTAPFFLHFLAQGTWSFGLAGCRLCHYVCGVSMYASVLLITAMSLDRSLAVA
RPFVSQKLRTKAMARRVLAGIWVLSFLLATPVLAYRTVVPWKTNMSLCFPRYPSEGHRAF
HLIFEAVTGFLLPFLAVVASYSDIGRRLQARRFRRSRRTGRLVVLIILTFAAFWLPYHVV
NLAEAGRALAGQAAGLGLVGKRLSLARNVLIALAFLSSSVNPVLYACAGGGLLRSAGVGF
VAKLLEGTGSEASSTRRGGSLGQTARSGPAALEPGPSESLTASSPLKLNELN
|
|
|
|---|
| BDBM50317625 |
|---|
| n/a |
|---|
| Name | BDBM50317625 |
|---|
| Synonyms: | 4-{2-(2-Carboxyethyl)-3-[6-([1,1',3,1'']terphenyl-5'-yloxy)-hexyl]phenoxy}butyric Acid | CHEMBL1099323 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C37H40O6 |
|---|
| Mol. Mass. | 580.7099 |
|---|
| SMILES | OC(=O)CCCOc1cccc(CCCCCCOc2cc(cc(c2)-c2ccccc2)-c2ccccc2)c1CCC(O)=O |
|---|
| Structure | 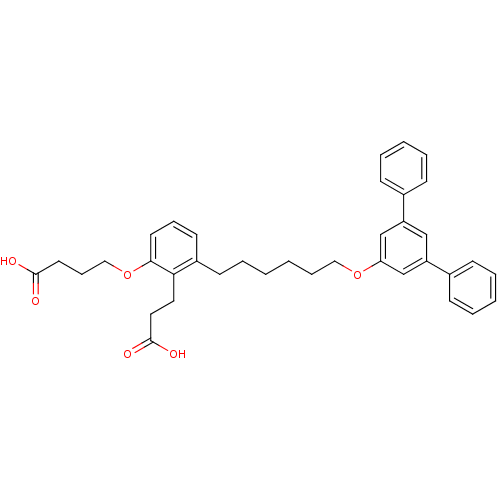
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Goodnow, RA; Hicks, A; Sidduri, A; Kowalczyk, A; Dominique, R; Qiao, Q; Lou, JP; Gillespie, P; Fotouhi, N; Tilley, J; Cohen, N; Choudhry, S; Cavallo, G; Tannu, SA; Ventre, JD; Lavelle, D; Tare, NS; Oh, H; Lamb, M; Kurylko, G; Hamid, R; Wright, MB; Pamidimukkala, A; Egan, T; Gubler, U; Hoffman, AF; Wei, X; Li, YL; O'Neil, J; Marcano, R; Pozzani, K; Molinaro, T; Santiago, J; Singer, L; Hargaden, M; Moore, D; Catala, AR; Chao, LC; Hermann, G; Venkat, R; Mancebo, H; Renzetti, LM Discovery of novel and potent leukotriene B4 receptor antagonists. Part 1. J Med Chem53:3502-16 (2010) [PubMed] Article
Goodnow, RA; Hicks, A; Sidduri, A; Kowalczyk, A; Dominique, R; Qiao, Q; Lou, JP; Gillespie, P; Fotouhi, N; Tilley, J; Cohen, N; Choudhry, S; Cavallo, G; Tannu, SA; Ventre, JD; Lavelle, D; Tare, NS; Oh, H; Lamb, M; Kurylko, G; Hamid, R; Wright, MB; Pamidimukkala, A; Egan, T; Gubler, U; Hoffman, AF; Wei, X; Li, YL; O'Neil, J; Marcano, R; Pozzani, K; Molinaro, T; Santiago, J; Singer, L; Hargaden, M; Moore, D; Catala, AR; Chao, LC; Hermann, G; Venkat, R; Mancebo, H; Renzetti, LM Discovery of novel and potent leukotriene B4 receptor antagonists. Part 1. J Med Chem53:3502-16 (2010) [PubMed] Article