| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Beta-2 adrenergic receptor |
|---|
| Ligand | BDBM50151720 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_632382 (CHEMBL1115587) |
|---|
| IC50 | 0.40±n/a nM |
|---|
| Citation |  Baur, F; Beattie, D; Beer, D; Bentley, D; Bradley, M; Bruce, I; Charlton, SJ; Cuenoud, B; Ernst, R; Fairhurst, RA; Faller, B; Farr, D; Keller, T; Fozard, JR; Fullerton, J; Garman, S; Hatto, J; Hayden, C; He, H; Howes, C; Janus, D; Jiang, Z; Lewis, C; Loeuillet-Ritzler, F; Moser, H; Reilly, J; Steward, A; Sykes, D; Tedaldi, L; Trifilieff, A; Tweed, M; Watson, S; Wissler, E; Wyss, D The identification of indacaterol as an ultralong-acting inhaled beta2-adrenoceptor agonist. J Med Chem53:3675-84 (2010) [PubMed] Article Baur, F; Beattie, D; Beer, D; Bentley, D; Bradley, M; Bruce, I; Charlton, SJ; Cuenoud, B; Ernst, R; Fairhurst, RA; Faller, B; Farr, D; Keller, T; Fozard, JR; Fullerton, J; Garman, S; Hatto, J; Hayden, C; He, H; Howes, C; Janus, D; Jiang, Z; Lewis, C; Loeuillet-Ritzler, F; Moser, H; Reilly, J; Steward, A; Sykes, D; Tedaldi, L; Trifilieff, A; Tweed, M; Watson, S; Wissler, E; Wyss, D The identification of indacaterol as an ultralong-acting inhaled beta2-adrenoceptor agonist. J Med Chem53:3675-84 (2010) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Beta-2 adrenergic receptor |
|---|
| Name: | Beta-2 adrenergic receptor |
|---|
| Synonyms: | ADRB2_CAVPO | Adrb2 | adrenergic Beta2 |
|---|
| Type: | Enzyme Catalytic Domain |
|---|
| Mol. Mass.: | 46993.24 |
|---|
| Organism: | GUINEA PIG |
|---|
| Description: | adrenergic Beta2 ADRB2 GUINEA PIG::Q8K4Z4 |
|---|
| Residue: | 418 |
|---|
| Sequence: | MGHLGNGSDFLLAPNASHAPDHNVTRERDEAWVVGMAIVMSLIVLAIVFGNVLVITAIAK
FERLQTVTNYFITSLACADLVMGLAVVPFGASHILMNMWTFGNFWCEFWTSIDVLCVTAS
IETLCVIAVDRYFAITSPFKYQSLLTKNKARVVILMVWVVSGLTSFLPIQMHWYRATHKD
AINCYAEETCCDFFTNQAYAIASSIVSFYLPLVVMVFVYSRVFQVAKKQLQKIDRSEGRF
HTQNLSQVEQDGRSGHGLRRSSKFYLKEHKALKTLGIIMGTFTLCWLPFFIVNIVHVIQD
NLIPKEVYILLNWVGYVNSAFNPLIYCRSPDFRIAFQELLCLRRSALKAYGNDCSSNSNG
KTDYTGEPNVCHQGQEKERELLCEDPPGTEDLVSCPGTVPSDSIDSQGRNYSTNDSLL
|
|
|
|---|
| BDBM50151720 |
|---|
| n/a |
|---|
| Name | BDBM50151720 |
|---|
| Synonyms: | ARFORMOTEROL TARTRATE | CHEMBL1363 | CHEMBL605993 | FORMOTEROL | FORMOTEROL FUMARATE | Foradil | N-(2-Hydroxy-5-{1-hydroxy-2-[2-(4-methoxy-phenyl)-1-methyl-ethylamino]-ethyl}-phenyl)-formamide | N-(2-hydroxy-5-((R)-1-hydroxy-2-((R)-1-(4-methoxyphenyl)propan-2-ylamino)ethyl)phenyl)formamide | Symbicort | cid_9912089 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C19H24N2O4 |
|---|
| Mol. Mass. | 344.4049 |
|---|
| SMILES | COc1ccc(C[C@@H](C)NC[C@H](O)c2ccc(O)c(NC=O)c2)cc1 |r| |
|---|
| Structure | 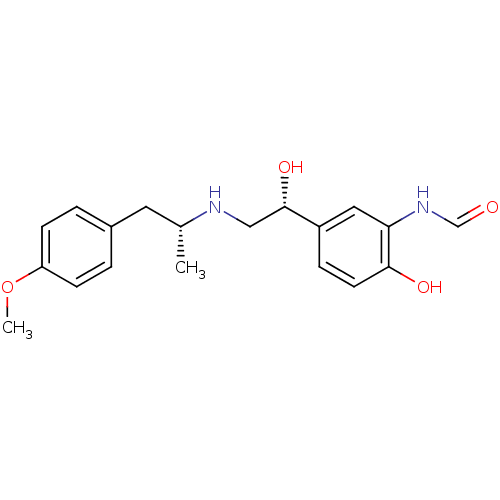
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Baur, F; Beattie, D; Beer, D; Bentley, D; Bradley, M; Bruce, I; Charlton, SJ; Cuenoud, B; Ernst, R; Fairhurst, RA; Faller, B; Farr, D; Keller, T; Fozard, JR; Fullerton, J; Garman, S; Hatto, J; Hayden, C; He, H; Howes, C; Janus, D; Jiang, Z; Lewis, C; Loeuillet-Ritzler, F; Moser, H; Reilly, J; Steward, A; Sykes, D; Tedaldi, L; Trifilieff, A; Tweed, M; Watson, S; Wissler, E; Wyss, D The identification of indacaterol as an ultralong-acting inhaled beta2-adrenoceptor agonist. J Med Chem53:3675-84 (2010) [PubMed] Article
Baur, F; Beattie, D; Beer, D; Bentley, D; Bradley, M; Bruce, I; Charlton, SJ; Cuenoud, B; Ernst, R; Fairhurst, RA; Faller, B; Farr, D; Keller, T; Fozard, JR; Fullerton, J; Garman, S; Hatto, J; Hayden, C; He, H; Howes, C; Janus, D; Jiang, Z; Lewis, C; Loeuillet-Ritzler, F; Moser, H; Reilly, J; Steward, A; Sykes, D; Tedaldi, L; Trifilieff, A; Tweed, M; Watson, S; Wissler, E; Wyss, D The identification of indacaterol as an ultralong-acting inhaled beta2-adrenoceptor agonist. J Med Chem53:3675-84 (2010) [PubMed] Article