| Reaction Details |
|---|
 | Report a problem with these data |
| Target | P2Y purinoceptor 4 |
|---|
| Ligand | BDBM50319141 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_635652 (CHEMBL1119544) |
|---|
| EC50 | 180±n/a nM |
|---|
| Citation |  Maruoka, H; Barrett, MO; Ko, H; Tosh, DK; Melman, A; Burianek, LE; Balasubramanian, R; Berk, B; Costanzi, S; Harden, TK; Jacobson, KA Pyrimidine ribonucleotides with enhanced selectivity as P2Y(6) receptor agonists: novel 4-alkyloxyimino, (S)-methanocarba, and 5'-triphosphate gamma-ester modifications. J Med Chem53:4488-501 (2010) [PubMed] Article Maruoka, H; Barrett, MO; Ko, H; Tosh, DK; Melman, A; Burianek, LE; Balasubramanian, R; Berk, B; Costanzi, S; Harden, TK; Jacobson, KA Pyrimidine ribonucleotides with enhanced selectivity as P2Y(6) receptor agonists: novel 4-alkyloxyimino, (S)-methanocarba, and 5'-triphosphate gamma-ester modifications. J Med Chem53:4488-501 (2010) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| P2Y purinoceptor 4 |
|---|
| Name: | P2Y purinoceptor 4 |
|---|
| Synonyms: | NRU | P2P | P2RY4 | P2RY4_HUMAN | P2Y purinoceptor 4 | P2Y4 | Pyrimidinergic receptor P2Y4 | UNR | Uridine nucleotide receptor |
|---|
| Type: | PROTEIN |
|---|
| Mol. Mass.: | 40977.17 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | ChEMBL_751027 |
|---|
| Residue: | 365 |
|---|
| Sequence: | MASTESSLLRSLGLSPGPGSSEVELDCWFDEDFKFILLPVSYAVVFVLGLGLNAPTLWLF
IFRLRPWDATATYMFHLALSDTLYVLSLPTLIYYYAAHNHWPFGTEICKFVRFLFYWNLY
CSVLFLTCISVHRYLGICHPLRALRWGRPRLAGLLCLAVWLVVAGCLVPNLFFVTTSNKG
TTVLCHDTTRPEEFDHYVHFSSAVMGLLFGVPCLVTLVCYGLMARRLYQPLPGSAQSSSR
LRSLRTIAVVLTVFAVCFVPFHITRTIYYLARLLEADCRVLNIVNVVYKVTRPLASANSC
LDPVLYLLTGDKYRRQLRQLCGGGKPQPRTAASSLALVSLPEDSSCRWAATPQDSSCSTP
RADRL
|
|
|
|---|
| BDBM50319141 |
|---|
| n/a |
|---|
| Name | BDBM50319141 |
|---|
| Synonyms: | CHEMBL1084295 | N4-Ethoxycytidine 5'-Diphosphate triethylAmmonium Salt |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C11H19N3O12P2 |
|---|
| Mol. Mass. | 447.229 |
|---|
| SMILES | CCONc1ccn([C@@H]2O[C@H](COP(O)(=O)OP(O)(O)=O)[C@@H](O)[C@H]2O)c(=O)n1 |r| |
|---|
| Structure | 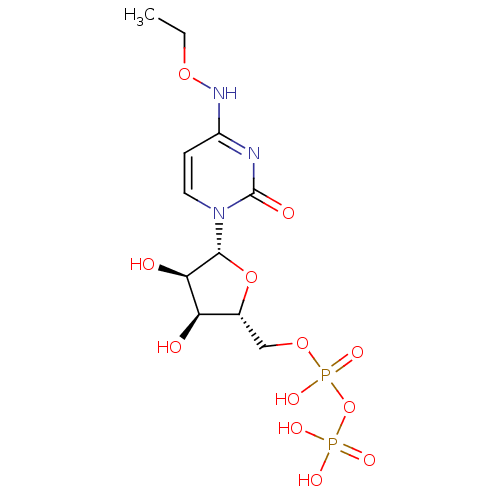
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Maruoka, H; Barrett, MO; Ko, H; Tosh, DK; Melman, A; Burianek, LE; Balasubramanian, R; Berk, B; Costanzi, S; Harden, TK; Jacobson, KA Pyrimidine ribonucleotides with enhanced selectivity as P2Y(6) receptor agonists: novel 4-alkyloxyimino, (S)-methanocarba, and 5'-triphosphate gamma-ester modifications. J Med Chem53:4488-501 (2010) [PubMed] Article
Maruoka, H; Barrett, MO; Ko, H; Tosh, DK; Melman, A; Burianek, LE; Balasubramanian, R; Berk, B; Costanzi, S; Harden, TK; Jacobson, KA Pyrimidine ribonucleotides with enhanced selectivity as P2Y(6) receptor agonists: novel 4-alkyloxyimino, (S)-methanocarba, and 5'-triphosphate gamma-ester modifications. J Med Chem53:4488-501 (2010) [PubMed] Article