| Reaction Details |
|---|
 | Report a problem with these data |
| Target | 5-hydroxytryptamine receptor 2A |
|---|
| Ligand | BDBM50319423 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_634153 (CHEMBL1119160) |
|---|
| IC50 | 5.2±n/a nM |
|---|
| Citation |  Xiong, Y; Teegarden, BR; Choi, JS; Strah-Pleynet, S; Decaire, M; Jayakumar, H; Dosa, PI; Casper, MD; Pham, L; Feichtinger, K; Ullman, B; Adams, J; Yuskin, D; Frazer, J; Morgan, M; Sadeque, A; Chen, W; Webb, RR; Connolly, DT; Semple, G; Al-Shamma, H Discovery and structure-activity relationship of 3-methoxy-N-(3-(1-methyl-1H-pyrazol-5-yl)-4-(2-morpholinoethoxy)phenyl)benzamide (APD791): a highly selective 5-hydroxytryptamine2A receptor inverse agonist for the treatment of arterial thrombosis. J Med Chem53:4412-21 (2010) [PubMed] Article Xiong, Y; Teegarden, BR; Choi, JS; Strah-Pleynet, S; Decaire, M; Jayakumar, H; Dosa, PI; Casper, MD; Pham, L; Feichtinger, K; Ullman, B; Adams, J; Yuskin, D; Frazer, J; Morgan, M; Sadeque, A; Chen, W; Webb, RR; Connolly, DT; Semple, G; Al-Shamma, H Discovery and structure-activity relationship of 3-methoxy-N-(3-(1-methyl-1H-pyrazol-5-yl)-4-(2-morpholinoethoxy)phenyl)benzamide (APD791): a highly selective 5-hydroxytryptamine2A receptor inverse agonist for the treatment of arterial thrombosis. J Med Chem53:4412-21 (2010) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| 5-hydroxytryptamine receptor 2A |
|---|
| Name: | 5-hydroxytryptamine receptor 2A |
|---|
| Synonyms: | 5-HT-2 | 5-HT-2A | 5-HT2A | 5-hydroxytryptamine receptor 2A (5-HT-2A) | 5-hydroxytryptamine receptor 2A (5HT-2A) | 5-hydroxytryptamine receptor 2A (5HT2A) | 5HT2A_HUMAN | HTR2 | HTR2A | Serotonin receptor 2A |
|---|
| Type: | undefined |
|---|
| Mol. Mass.: | 52607.65 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | P28223 |
|---|
| Residue: | 471 |
|---|
| Sequence: | MDILCEENTSLSSTTNSLMQLNDDTRLYSNDFNSGEANTSDAFNWTVDSENRTNLSCEGC
LSPSCLSLLHLQEKNWSALLTAVVIILTIAGNILVIMAVSLEKKLQNATNYFLMSLAIAD
MLLGFLVMPVSMLTILYGYRWPLPSKLCAVWIYLDVLFSTASIMHLCAISLDRYVAIQNP
IHHSRFNSRTKAFLKIIAVWTISVGISMPIPVFGLQDDSKVFKEGSCLLADDNFVLIGSF
VSFFIPLTIMVITYFLTIKSLQKEATLCVSDLGTRAKLASFSFLPQSSLSSEKLFQRSIH
REPGSYTGRRTMQSISNEQKACKVLGIVFFLFVVMWCPFFITNIMAVICKESCNEDVIGA
LLNVFVWIGYLSSAVNPLVYTLFNKTYRSAFSRYIQCQYKENKKPLQLILVNTIPALAYK
SSQLQMGQKKNSKQDAKTTDNDCSMVALGKQHSEEASKDNSDGVNEKVSCV
|
|
|
|---|
| BDBM50319423 |
|---|
| n/a |
|---|
| Name | BDBM50319423 |
|---|
| Synonyms: | 3-Methoxy-N-[3-(2-methyl-2H-pyrazol-3-yl)-4-(2-morpholin-4-yl-ethoxy)-phenyl]-benzamide | CHEMBL1084617 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C24H28N4O4 |
|---|
| Mol. Mass. | 436.5035 |
|---|
| SMILES | COc1cccc(c1)C(=O)Nc1ccc(OCCN2CCOCC2)c(c1)-c1ccnn1C |
|---|
| Structure | 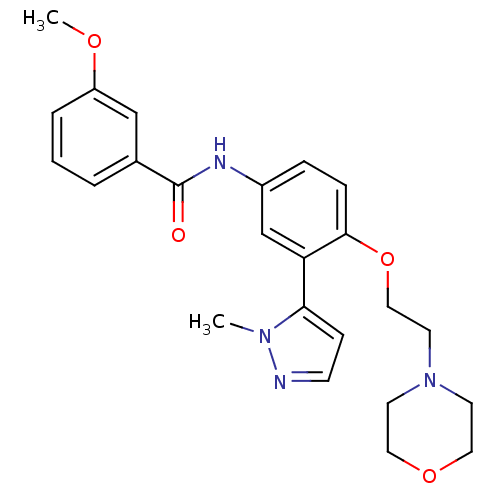
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Xiong, Y; Teegarden, BR; Choi, JS; Strah-Pleynet, S; Decaire, M; Jayakumar, H; Dosa, PI; Casper, MD; Pham, L; Feichtinger, K; Ullman, B; Adams, J; Yuskin, D; Frazer, J; Morgan, M; Sadeque, A; Chen, W; Webb, RR; Connolly, DT; Semple, G; Al-Shamma, H Discovery and structure-activity relationship of 3-methoxy-N-(3-(1-methyl-1H-pyrazol-5-yl)-4-(2-morpholinoethoxy)phenyl)benzamide (APD791): a highly selective 5-hydroxytryptamine2A receptor inverse agonist for the treatment of arterial thrombosis. J Med Chem53:4412-21 (2010) [PubMed] Article
Xiong, Y; Teegarden, BR; Choi, JS; Strah-Pleynet, S; Decaire, M; Jayakumar, H; Dosa, PI; Casper, MD; Pham, L; Feichtinger, K; Ullman, B; Adams, J; Yuskin, D; Frazer, J; Morgan, M; Sadeque, A; Chen, W; Webb, RR; Connolly, DT; Semple, G; Al-Shamma, H Discovery and structure-activity relationship of 3-methoxy-N-(3-(1-methyl-1H-pyrazol-5-yl)-4-(2-morpholinoethoxy)phenyl)benzamide (APD791): a highly selective 5-hydroxytryptamine2A receptor inverse agonist for the treatment of arterial thrombosis. J Med Chem53:4412-21 (2010) [PubMed] Article