

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
| Reaction Details | |||
|---|---|---|---|
 | Report a problem with these data | ||
| Target | Serine/threonine-protein kinase mTOR | ||
| Ligand | BDBM50320095 | ||
| Substrate/Competitor | n/a | ||
| Meas. Tech. | ChEMBL_635345 (CHEMBL1119658) | ||
| IC50 | 2.4±n/a nM | ||
| Citation |  Zhang, N; Ayral-Kaloustian, S; Anderson, JT; Nguyen, T; Das, S; Venkatesan, AM; Brooijmans, N; Lucas, J; Yu, K; Hollander, I; Mallon, R 5-ureidobenzofuranone indoles as potent and efficacious inhibitors of PI3 kinase-alpha and mTOR for the treatment of breast cancer. Bioorg Med Chem Lett20:3526-9 (2010) [PubMed] Article Zhang, N; Ayral-Kaloustian, S; Anderson, JT; Nguyen, T; Das, S; Venkatesan, AM; Brooijmans, N; Lucas, J; Yu, K; Hollander, I; Mallon, R 5-ureidobenzofuranone indoles as potent and efficacious inhibitors of PI3 kinase-alpha and mTOR for the treatment of breast cancer. Bioorg Med Chem Lett20:3526-9 (2010) [PubMed] Article | ||
| More Info.: | Get all data from this article, Assay Method | ||
| Serine/threonine-protein kinase mTOR | |||
| Name: | Serine/threonine-protein kinase mTOR | ||
| Synonyms: | FK506-binding protein 12-rapamycin complex-associated protein 1 | FKBP12-rapamycin complex-associated protein | FRAP | FRAP 1 (mTOR) | FRAP1 | FRAP2 | MTOR | MTOR_HUMAN | Mammalian Target of Rapamycin (mTOR) | P42345 | RAFT1 | RAPT1 | Rapamycin and FKBP12 target 1 | Rapamycin target protein | Serine/threonine-protein kinase (mTOR) | mTORC2 | ||
| Type: | Rapamycin target protein | ||
| Mol. Mass.: | 288917.12 | ||
| Organism: | Homo sapiens (Human) | ||
| Description: | P42345 | ||
| Residue: | 2549 | ||
| Sequence: |
| ||
| BDBM50320095 | |||
| n/a | |||
| Name | BDBM50320095 | ||
| Synonyms: | 1-(2-((7-fluoro-5-methoxy-2-(1,3,5-trimethyl-1H-pyrazol-4-yl)-1H-indol-3-yl)methylene)-3-oxo-2,3-dihydrobenzofuran-5-yl)-3-methylurea | CHEMBL1083305 | ||
| Type | Small organic molecule | ||
| Emp. Form. | C26H24FN5O4 | ||
| Mol. Mass. | 489.4983 | ||
| SMILES | CNC(=O)Nc1ccc2O\C(=C\c3c([nH]c4c(F)cc(OC)cc34)-c3c(C)nn(C)c3C)C(=O)c2c1 |(15.41,-12.32,;16.73,-11.54,;18.07,-12.3,;19.4,-11.53,;18.07,-13.84,;19.41,-14.61,;20.75,-13.83,;22.07,-14.6,;22.07,-16.14,;23.22,-17.16,;22.59,-18.56,;23.36,-19.9,;22.76,-21.32,;23.67,-22.57,;22.77,-23.82,;21.3,-23.34,;19.96,-24.12,;19.97,-25.65,;18.63,-23.35,;18.63,-21.81,;17.29,-21.03,;17.29,-19.49,;19.96,-21.03,;21.3,-21.8,;25.21,-22.57,;26.11,-23.81,;25.63,-25.28,;27.57,-23.34,;27.57,-21.8,;28.82,-20.89,;26.11,-21.32,;25.63,-19.85,;21.06,-18.41,;20.03,-19.56,;20.75,-16.9,;19.42,-16.14,)| | ||
| Structure | 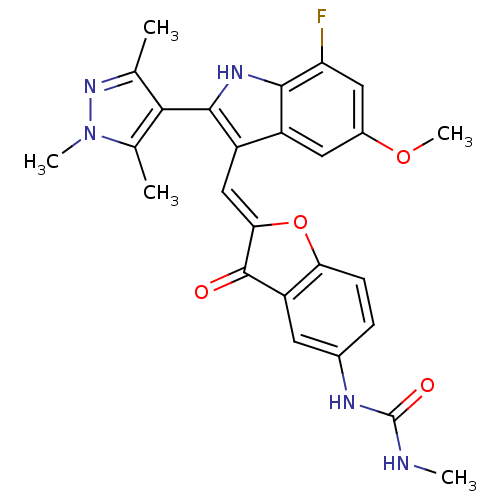
| ||