| Reaction Details |
|---|
 | Report a problem with these data |
| Target | 72 kDa type IV collagenase |
|---|
| Ligand | BDBM50320444 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_634117 (CHEMBL1119124) |
|---|
| IC50 | 6350±n/a nM |
|---|
| Citation |  Kolodziej, SA; Hockerman, SL; DeCrescenzo, GA; McDonald, JJ; Mischke, DA; Munie, GE; Fletcher, TR; Stehle, N; Swearingen, C; Becker, DP MMP-13 selective isonipecotamide alpha-sulfone hydroxamates. Bioorg Med Chem Lett20:3561-4 (2010) [PubMed] Article Kolodziej, SA; Hockerman, SL; DeCrescenzo, GA; McDonald, JJ; Mischke, DA; Munie, GE; Fletcher, TR; Stehle, N; Swearingen, C; Becker, DP MMP-13 selective isonipecotamide alpha-sulfone hydroxamates. Bioorg Med Chem Lett20:3561-4 (2010) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| 72 kDa type IV collagenase |
|---|
| Name: | 72 kDa type IV collagenase |
|---|
| Synonyms: | 72 kDa gelatinase | 72 kDa type IV collagenase precursor | CLG4A | Gelatinase A | Gelatinase A (MMP-2) | MMP2 | MMP2_HUMAN | Matrix metalloproteinase-2 | Matrix metalloproteinase-2 (MMP 2) | Matrix metalloproteinase-2 (MMP2) | Matrix metalloproteinases 2 (MMP-2) | TBE-1 |
|---|
| Type: | Enzyme |
|---|
| Mol. Mass.: | 73870.36 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | P08253 |
|---|
| Residue: | 660 |
|---|
| Sequence: | MEALMARGALTGPLRALCLLGCLLSHAAAAPSPIIKFPGDVAPKTDKELAVQYLNTFYGC
PKESCNLFVLKDTLKKMQKFFGLPQTGDLDQNTIETMRKPRCGNPDVANYNFFPRKPKWD
KNQITYRIIGYTPDLDPETVDDAFARAFQVWSDVTPLRFSRIHDGEADIMINFGRWEHGD
GYPFDGKDGLLAHAFAPGTGVGGDSHFDDDELWTLGEGQVVRVKYGNADGEYCKFPFLFN
GKEYNSCTDTGRSDGFLWCSTTYNFEKDGKYGFCPHEALFTMGGNAEGQPCKFPFRFQGT
SYDSCTTEGRTDGYRWCGTTEDYDRDKKYGFCPETAMSTVGGNSEGAPCVFPFTFLGNKY
ESCTSAGRSDGKMWCATTANYDDDRKWGFCPDQGYSLFLVAAHEFGHAMGLEHSQDPGAL
MAPIYTYTKNFRLSQDDIKGIQELYGASPDIDLGTGPTPTLGPVTPEICKQDIVFDGIAQ
IRGEIFFFKDRFIWRTVTPRDKPMGPLLVATFWPELPEKIDAVYEAPQEEKAVFFAGNEY
WIYSASTLERGYPKPLTSLGLPPDVQRVDAAFNWSKNKKTYIFAGDKFWRYNEVKKKMDP
GFPKLIADAWNAIPDNLDAVVDLQGGGHSYFFKGAYYLKLENQSLKSVKFGSIKSDWLGC
|
|
|
|---|
| BDBM50320444 |
|---|
| n/a |
|---|
| Name | BDBM50320444 |
|---|
| Synonyms: | 1-cyclopropyl-4-(4-(4-(4-(2,3-dimethylphenyl)piperazine-1-carbonyl)piperidin-1-yl)phenylsulfonyl)-N-hydroxypiperidine-4-carboxamide | CHEMBL1082318 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C33H45N5O5S |
|---|
| Mol. Mass. | 623.806 |
|---|
| SMILES | Cc1cccc(N2CCN(CC2)C(=O)C2CCN(CC2)c2ccc(cc2)S(=O)(=O)C2(CCN(CC2)C2CC2)C(=O)NO)c1C |
|---|
| Structure | 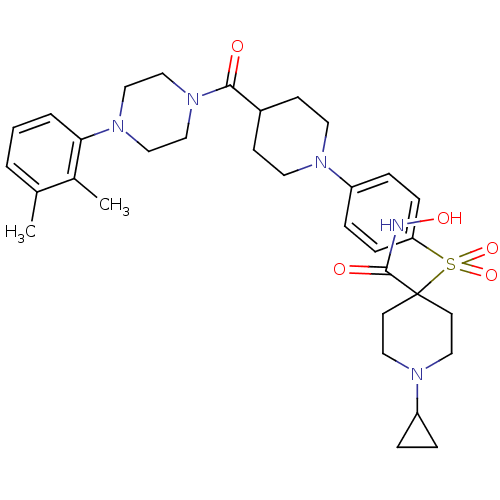
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Kolodziej, SA; Hockerman, SL; DeCrescenzo, GA; McDonald, JJ; Mischke, DA; Munie, GE; Fletcher, TR; Stehle, N; Swearingen, C; Becker, DP MMP-13 selective isonipecotamide alpha-sulfone hydroxamates. Bioorg Med Chem Lett20:3561-4 (2010) [PubMed] Article
Kolodziej, SA; Hockerman, SL; DeCrescenzo, GA; McDonald, JJ; Mischke, DA; Munie, GE; Fletcher, TR; Stehle, N; Swearingen, C; Becker, DP MMP-13 selective isonipecotamide alpha-sulfone hydroxamates. Bioorg Med Chem Lett20:3561-4 (2010) [PubMed] Article