| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Neuropeptide Y receptor type 2 |
|---|
| Ligand | BDBM50323040 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_643625 (CHEMBL1212489) |
|---|
| IC50 | >10000±n/a nM |
|---|
| Citation |  Gattinoni, S; Simone, CD; Dallavalle, S; Fezza, F; Nannei, R; Battista, N; Minetti, P; Quattrociocchi, G; Caprioli, A; Borsini, F; Cabri, W; Penco, S; Merlini, L; Maccarrone, M A new group of oxime carbamates as reversible inhibitors of fatty acid amide hydrolase. Bioorg Med Chem Lett20:4406-11 (2010) [PubMed] Article Gattinoni, S; Simone, CD; Dallavalle, S; Fezza, F; Nannei, R; Battista, N; Minetti, P; Quattrociocchi, G; Caprioli, A; Borsini, F; Cabri, W; Penco, S; Merlini, L; Maccarrone, M A new group of oxime carbamates as reversible inhibitors of fatty acid amide hydrolase. Bioorg Med Chem Lett20:4406-11 (2010) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Neuropeptide Y receptor type 2 |
|---|
| Name: | Neuropeptide Y receptor type 2 |
|---|
| Synonyms: | NPY-Y2 | NPY-Y2 receptor | NPY2-R | NPY2R | NPY2R_HUMAN | Neuropeptide Y receptor type 2 | Y2 receptor | neuropeptide Y receptor Y2 |
|---|
| Type: | Enzyme Catalytic Domain |
|---|
| Mol. Mass.: | 42734.65 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | NPY-Y2 NPY2R HUMAN::P49146 |
|---|
| Residue: | 381 |
|---|
| Sequence: | MGPIGAEADENQTVEEMKVEQYGPQTTPRGELVPDPEPELIDSTKLIEVQVVLILAYCSI
ILLGVIGNSLVIHVVIKFKSMRTVTNFFIANLAVADLLVNTLCLPFTLTYTLMGEWKMGP
VLCHLVPYAQGLAVQVSTITLTVIALDRHRCIVYHLESKISKRISFLIIGLAWGISALLA
SPLAIFREYSLIEIIPDFEIVACTEKWPGEEKSIYGTVYSLSSLLILYVLPLGIISFSYT
RIWSKLKNHVSPGAANDHYHQRRQKTTKMLVCVVVVFAVSWLPLHAFQLAVDIDSQVLDL
KEYKLIFTVFHIIAMCSTFANPLLYGWMNSNYRKAFLSAFRCEQRLDAIHSEVSVTFKAK
KNLEVRKNSGPNDSFTEATNV
|
|
|
|---|
| BDBM50323040 |
|---|
| n/a |
|---|
| Name | BDBM50323040 |
|---|
| Synonyms: | 1-(3-Thiophen-2-yl-phenyl)-ethanone, O-(phenylaminocarbonyl)oxime | CHEMBL1210154 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C19H16N2O2S |
|---|
| Mol. Mass. | 336.408 |
|---|
| SMILES | CC(=NOC(=O)Nc1ccccc1)c1cccc(c1)-c1cccs1 |w:2.2| |
|---|
| Structure | 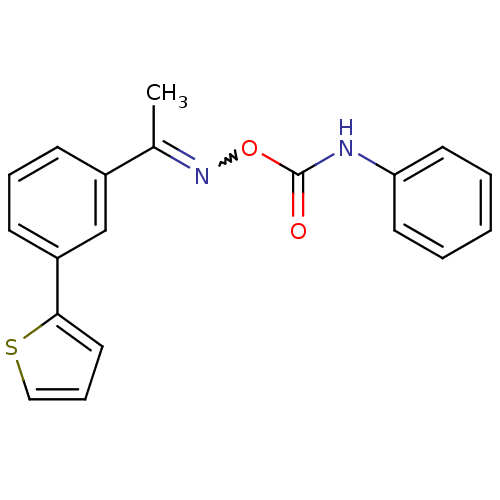
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Gattinoni, S; Simone, CD; Dallavalle, S; Fezza, F; Nannei, R; Battista, N; Minetti, P; Quattrociocchi, G; Caprioli, A; Borsini, F; Cabri, W; Penco, S; Merlini, L; Maccarrone, M A new group of oxime carbamates as reversible inhibitors of fatty acid amide hydrolase. Bioorg Med Chem Lett20:4406-11 (2010) [PubMed] Article
Gattinoni, S; Simone, CD; Dallavalle, S; Fezza, F; Nannei, R; Battista, N; Minetti, P; Quattrociocchi, G; Caprioli, A; Borsini, F; Cabri, W; Penco, S; Merlini, L; Maccarrone, M A new group of oxime carbamates as reversible inhibitors of fatty acid amide hydrolase. Bioorg Med Chem Lett20:4406-11 (2010) [PubMed] Article