| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Protein kinase C beta type |
|---|
| Ligand | BDBM50327943 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_665131 (CHEMBL1260394) |
|---|
| Ki | 0.450000±n/a nM |
|---|
| Citation |  Yanagita, RC; Kamachi, H; Tanaka, K; Murakami, A; Nakagawa, Y; Tokuda, H; Nagai, H; Irie, K Role of the phenolic hydroxyl group in the biological activities of simplified analogue of aplysiatoxin with antiproliferative activity. Bioorg Med Chem Lett20:6064-6 (2010) [PubMed] Article Yanagita, RC; Kamachi, H; Tanaka, K; Murakami, A; Nakagawa, Y; Tokuda, H; Nagai, H; Irie, K Role of the phenolic hydroxyl group in the biological activities of simplified analogue of aplysiatoxin with antiproliferative activity. Bioorg Med Chem Lett20:6064-6 (2010) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Protein kinase C beta type |
|---|
| Name: | Protein kinase C beta type |
|---|
| Synonyms: | KPCB_HUMAN | PKC alpha and beta-2 | PKC beta | PKC beta isoform 2 | PKCB | PRKCB | PRKCB1 | Protein Kinase C, beta II | Protein kinase C beta type | Protein kinase C beta type II (PRKCB2) |
|---|
| Type: | Enzyme |
|---|
| Mol. Mass.: | 76873.67 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | The recombinant human PKC enzymes were
produced using a baculovirus expression system in SF9 cells
|
|---|
| Residue: | 671 |
|---|
| Sequence: | MADPAAGPPPSEGEESTVRFARKGALRQKNVHEVKNHKFTARFFKQPTFCSHCTDFIWGF
GKQGFQCQVCCFVVHKRCHEFVTFSCPGADKGPASDDPRSKHKFKIHTYSSPTFCDHCGS
LLYGLIHQGMKCDTCMMNVHKRCVMNVPSLCGTDHTERRGRIYIQAHIDRDVLIVLVRDA
KNLVPMDPNGLSDPYVKLKLIPDPKSESKQKTKTIKCSLNPEWNETFRFQLKESDKDRRL
SVEIWDWDLTSRNDFMGSLSFGISELQKASVDGWFKLLSQEEGEYFNVPVPPEGSEANEE
LRQKFERAKISQGTKVPEEKTTNTVSKFDNNGNRDRMKLTDFNFLMVLGKGSFGKVMLSE
RKGTDELYAVKILKKDVVIQDDDVECTMVEKRVLALPGKPPFLTQLHSCFQTMDRLYFVM
EYVNGGDLMYHIQQVGRFKEPHAVFYAAEIAIGLFFLQSKGIIYRDLKLDNVMLDSEGHI
KIADFGMCKENIWDGVTTKTFCGTPDYIAPEIIAYQPYGKSVDWWAFGVLLYEMLAGQAP
FEGEDEDELFQSIMEHNVAYPKSMSKEAVAICKGLMTKHPGKRLGCGPEGERDIKEHAFF
RYIDWEKLERKEIQPPYKPKARDKRDTSNFDKEFTRQPVELTPTDKLFIMNLDQNEFAGF
SYTNPEFVINV
|
|
|
|---|
| BDBM50327943 |
|---|
| n/a |
|---|
| Name | BDBM50327943 |
|---|
| Synonyms: | Aplysiatoxin | CHEMBL1256416 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C32H47BrO10 |
|---|
| Mol. Mass. | 671.614 |
|---|
| SMILES | CO[C@@H](CC[C@H](C)[C@H]1O[C@@]23C[C@H](OC(=O)C[C@@H](OC(=O)C[C@](O)(O2)[C@H](C)CC3(C)C)[C@@H](C)O)[C@@H]1C)c1cc(O)ccc1Br |r| |
|---|
| Structure | 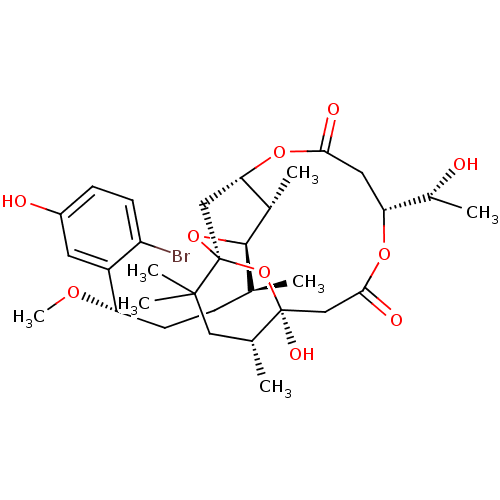
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Yanagita, RC; Kamachi, H; Tanaka, K; Murakami, A; Nakagawa, Y; Tokuda, H; Nagai, H; Irie, K Role of the phenolic hydroxyl group in the biological activities of simplified analogue of aplysiatoxin with antiproliferative activity. Bioorg Med Chem Lett20:6064-6 (2010) [PubMed] Article
Yanagita, RC; Kamachi, H; Tanaka, K; Murakami, A; Nakagawa, Y; Tokuda, H; Nagai, H; Irie, K Role of the phenolic hydroxyl group in the biological activities of simplified analogue of aplysiatoxin with antiproliferative activity. Bioorg Med Chem Lett20:6064-6 (2010) [PubMed] Article