| Reaction Details |
|---|
 | Report a problem with these data |
| Target | 11-beta-hydroxysteroid dehydrogenase type 2 |
|---|
| Ligand | BDBM50329193 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_674863 (CHEMBL1272436) |
|---|
| IC50 | 61.1±n/a nM |
|---|
| Citation |  Stanetty, C; Czollner, L; Koller, I; Shah, P; Gaware, R; Cunha, TD; Odermatt, A; Jordis, U; Kosma, P; Classen-Houben, D Synthesis of novel 3-amino and 29-hydroxamic acid derivatives of glycyrrhetinic acid as selective 11ß-hydroxysteroid dehydrogenase 2 inhibitors. Bioorg Med Chem18:7522-41 (2010) [PubMed] Article Stanetty, C; Czollner, L; Koller, I; Shah, P; Gaware, R; Cunha, TD; Odermatt, A; Jordis, U; Kosma, P; Classen-Houben, D Synthesis of novel 3-amino and 29-hydroxamic acid derivatives of glycyrrhetinic acid as selective 11ß-hydroxysteroid dehydrogenase 2 inhibitors. Bioorg Med Chem18:7522-41 (2010) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| 11-beta-hydroxysteroid dehydrogenase type 2 |
|---|
| Name: | 11-beta-hydroxysteroid dehydrogenase type 2 |
|---|
| Synonyms: | 11-DH2 | 11-beta-HSD2 | 11-beta-Hydroxysteroid Dehydrogenase 2 (11-beta-HSD2) | 11-beta-hydroxysteroid dehydrogenase | 11-beta-hydroxysteroid dehydrogenase 2 | 11-beta-hydroxysteroid dehydrogenase type 2 | 11-beta-hydroxysteroid dehydrogenase type 2 (11-beta-HSD2) | Corticosteroid 11-beta-dehydrogenase isozyme 2 | DHI2_HUMAN | HSD11B2 | HSD11K | NAD-dependent 11-beta-hydroxysteroid dehydrogenase | SDR9C3 |
|---|
| Type: | Enzyme |
|---|
| Mol. Mass.: | 44141.72 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | Purified recombinant human 11beta-HSD2. |
|---|
| Residue: | 405 |
|---|
| Sequence: | MERWPWPSGGAWLLVAARALLQLLRSDLRLGRPLLAALALLAALDWLCQRLLPPPAALAV
LAAAGWIALSRLARPQRLPVATRAVLITGCDSGFGKETAKKLDSMGFTVLATVLELNSPG
AIELRTCCSPRLRLLQMDLTKPGDISRVLEFTKAHTTSTGLWGLVNNAGHNEVVADAELS
PVATFRSCMEVNFFGALELTKGLLPLLRSSRGRIVTVGSPAGDMPYPCLGAYGTSKAAVA
LLMDTFSCELLPWGVKVSIIQPGCFKTESVRNVGQWEKRKQLLLANLPQELLQAYGKDYI
EHLHGQFLHSLRLAMSDLTPVVDAITDALLAARPRRRYYPGQGLGLMYFIHYYLPEGLRR
RFLQAFFISHCLPRALQPGQPGTTPPQDAAQDPNLSPGPSPAVAR
|
|
|
|---|
| BDBM50329193 |
|---|
| n/a |
|---|
| Name | BDBM50329193 |
|---|
| Synonyms: | (3beta,18beta,20beta)-3-Acetoxy-N-hydroxy-11-oxo-olean-12-en-29-amide | CHEMBL1270688 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C32H49NO5 |
|---|
| Mol. Mass. | 527.7352 |
|---|
| SMILES | CC(=O)O[C@H]1CC[C@@]2(C)[C@@H](CC[C@]3(C)[C@@H]2C(=O)C=C2[C@@H]4C[C@](C)(CC[C@]4(C)CC[C@@]32C)C(=O)NO)C1(C)C |r,t:18| |
|---|
| Structure | 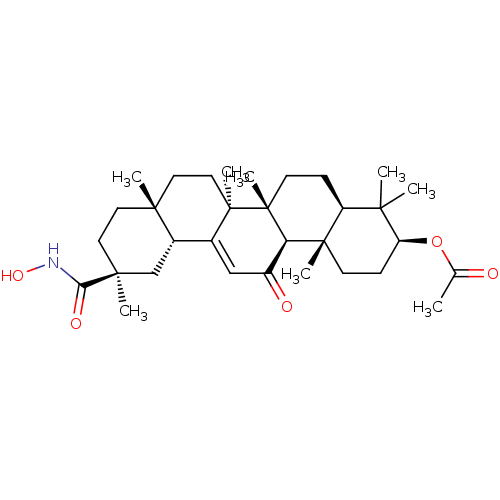
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Stanetty, C; Czollner, L; Koller, I; Shah, P; Gaware, R; Cunha, TD; Odermatt, A; Jordis, U; Kosma, P; Classen-Houben, D Synthesis of novel 3-amino and 29-hydroxamic acid derivatives of glycyrrhetinic acid as selective 11ß-hydroxysteroid dehydrogenase 2 inhibitors. Bioorg Med Chem18:7522-41 (2010) [PubMed] Article
Stanetty, C; Czollner, L; Koller, I; Shah, P; Gaware, R; Cunha, TD; Odermatt, A; Jordis, U; Kosma, P; Classen-Houben, D Synthesis of novel 3-amino and 29-hydroxamic acid derivatives of glycyrrhetinic acid as selective 11ß-hydroxysteroid dehydrogenase 2 inhibitors. Bioorg Med Chem18:7522-41 (2010) [PubMed] Article