| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Cytochrome P450 3A4 |
|---|
| Ligand | BDBM50330752 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_685219 (CHEMBL1287107) |
|---|
| IC50 | >10000±n/a nM |
|---|
| Citation |  Gianotti, M; Botta, M; Brough, S; Carletti, R; Castiglioni, E; Corti, C; Dal-Cin, M; Delle Fratte, S; Korajac, D; Lovric, M; Merlo, G; Mesic, M; Pavone, F; Piccoli, L; Rast, S; Roscic, M; Sava, A; Smehil, M; Stasi, L; Togninelli, A; Wigglesworth, MJ Novel spirotetracyclic zwitterionic dual H(1)/5-HT(2A) receptor antagonists for the treatment of sleep disorders. J Med Chem53:7778-95 (2010) [PubMed] Article Gianotti, M; Botta, M; Brough, S; Carletti, R; Castiglioni, E; Corti, C; Dal-Cin, M; Delle Fratte, S; Korajac, D; Lovric, M; Merlo, G; Mesic, M; Pavone, F; Piccoli, L; Rast, S; Roscic, M; Sava, A; Smehil, M; Stasi, L; Togninelli, A; Wigglesworth, MJ Novel spirotetracyclic zwitterionic dual H(1)/5-HT(2A) receptor antagonists for the treatment of sleep disorders. J Med Chem53:7778-95 (2010) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Cytochrome P450 3A4 |
|---|
| Name: | Cytochrome P450 3A4 |
|---|
| Synonyms: | Albendazole monooxygenase | Albendazole sulfoxidase | CP3A4_HUMAN | CYP3A3 | CYP3A4 | CYPIIIA3 | CYPIIIA4 | Cytochrome P450 3A3 | Cytochrome P450 3A4 (CYP3A4) | Cytochrome P450 HLp | Nifedipine oxidase | Quinine 3-monooxygenase | Taurochenodeoxycholate 6-alpha-hydroxylase |
|---|
| Type: | Enzyme |
|---|
| Mol. Mass.: | 57349.57 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | n/a |
|---|
| Residue: | 503 |
|---|
| Sequence: | MALIPDLAMETWLLLAVSLVLLYLYGTHSHGLFKKLGIPGPTPLPFLGNILSYHKGFCMF
DMECHKKYGKVWGFYDGQQPVLAITDPDMIKTVLVKECYSVFTNRRPFGPVGFMKSAISI
AEDEEWKRLRSLLSPTFTSGKLKEMVPIIAQYGDVLVRNLRREAETGKPVTLKDVFGAYS
MDVITSTSFGVNIDSLNNPQDPFVENTKKLLRFDFLDPFFLSITVFPFLIPILEVLNICV
FPREVTNFLRKSVKRMKESRLEDTQKHRVDFLQLMIDSQNSKETESHKALSDLELVAQSI
IFIFAGYETTSSVLSFIMYELATHPDVQQKLQEEIDAVLPNKAPPTYDTVLQMEYLDMVV
NETLRLFPIAMRLERVCKKDVEINGMFIPKGVVVMIPSYALHRDPKYWTEPEKFLPERFS
KKNKDNIDPYIYTPFGSGPRNCIGMRFALMNMKLALIRVLQNFSFKPCKETQIPLKLSLG
GLLQPEKPVVLKVESRDGTVSGA
|
|
|
|---|
| BDBM50330752 |
|---|
| n/a |
|---|
| Name | BDBM50330752 |
|---|
| Synonyms: | 3-((1S)-5',11'-Dihydrospiro[cyclopentane-1,10'-dibenzo[a,d]-cyclohepten]-3-yl)-3-azabicyclo[3.1.0]hexane-1-carboxylic Acid | CHEMBL1276947 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C25H27NO2 |
|---|
| Mol. Mass. | 373.4874 |
|---|
| SMILES | OC(=O)[C@]12C[C@H]1CN(C2)C1CC[C@@]2(C1)Cc1ccccc1Cc1ccccc21 |r| |
|---|
| Structure | 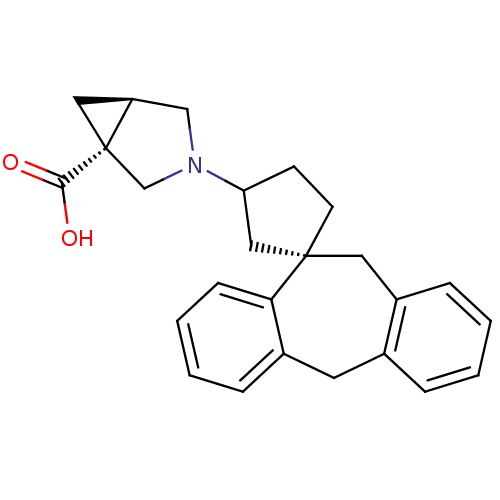
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Gianotti, M; Botta, M; Brough, S; Carletti, R; Castiglioni, E; Corti, C; Dal-Cin, M; Delle Fratte, S; Korajac, D; Lovric, M; Merlo, G; Mesic, M; Pavone, F; Piccoli, L; Rast, S; Roscic, M; Sava, A; Smehil, M; Stasi, L; Togninelli, A; Wigglesworth, MJ Novel spirotetracyclic zwitterionic dual H(1)/5-HT(2A) receptor antagonists for the treatment of sleep disorders. J Med Chem53:7778-95 (2010) [PubMed] Article
Gianotti, M; Botta, M; Brough, S; Carletti, R; Castiglioni, E; Corti, C; Dal-Cin, M; Delle Fratte, S; Korajac, D; Lovric, M; Merlo, G; Mesic, M; Pavone, F; Piccoli, L; Rast, S; Roscic, M; Sava, A; Smehil, M; Stasi, L; Togninelli, A; Wigglesworth, MJ Novel spirotetracyclic zwitterionic dual H(1)/5-HT(2A) receptor antagonists for the treatment of sleep disorders. J Med Chem53:7778-95 (2010) [PubMed] Article