| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Cathepsin D |
|---|
| Ligand | BDBM50333943 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_700285 (CHEMBL1647503) |
|---|
| Ki | 3.3±n/a nM |
|---|
| Citation |  Sund, C; Belda, O; Wiktelius, D; Sahlberg, C; Vrang, L; Sedig, S; Hamelink, E; Henderson, I; Agback, T; Jansson, K; Borkakoti, N; Derbyshire, D; Eneroth, A; Samuelsson, B Design and synthesis of potent macrocyclic renin inhibitors. Bioorg Med Chem Lett21:358-62 (2010) [PubMed] Article Sund, C; Belda, O; Wiktelius, D; Sahlberg, C; Vrang, L; Sedig, S; Hamelink, E; Henderson, I; Agback, T; Jansson, K; Borkakoti, N; Derbyshire, D; Eneroth, A; Samuelsson, B Design and synthesis of potent macrocyclic renin inhibitors. Bioorg Med Chem Lett21:358-62 (2010) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Cathepsin D |
|---|
| Name: | Cathepsin D |
|---|
| Synonyms: | CATD_HUMAN | CPSD | CTSD | Cathepsin D [Precursor] | Cathepsin D heavy chain | Cathepsin D light chain | Cathepsin D precursor |
|---|
| Type: | Enzyme |
|---|
| Mol. Mass.: | 44551.72 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | Human proCathepsin D (SwissProt accession number P07339) was expressed in Sf9 cells, purified, and autoactivated. |
|---|
| Residue: | 412 |
|---|
| Sequence: | MQPSSLLPLALCLLAAPASALVRIPLHKFTSIRRTMSEVGGSVEDLIAKGPVSKYSQAVP
AVTEGPIPEVLKNYMDAQYYGEIGIGTPPQCFTVVFDTGSSNLWVPSIHCKLLDIACWIH
HKYNSDKSSTYVKNGTSFDIHYGSGSLSGYLSQDTVSVPCQSASSASALGGVKVERQVFG
EATKQPGITFIAAKFDGILGMAYPRISVNNVLPVFDNLMQQKLVDQNIFSFYLSRDPDAQ
PGGELMLGGTDSKYYKGSLSYLNVTRKAYWQVHLDQVEVASGLTLCKEGCEAIVDTGTSL
MVGPVDEVRELQKAIGAVPLIQGEYMIPCEKVSTLPAITLKLGGKGYKLSPEDYTLKVSQ
AGKTLCLSGFMGMDIPPPSGPLWILGDVFIGRYYTVFDRDNNRVGFAEAARL
|
|
|
|---|
| BDBM50333943 |
|---|
| n/a |
|---|
| Name | BDBM50333943 |
|---|
| Synonyms: | CHEMBL1644461 | N1-((2S,3S,5S)-5-((S)-1-(benzylamino)-3-methyl-1-oxobutan-2-ylcarbamoyl)-1-(benzyloxy)-3-hydroxy-6-methylheptan-2-yl)-5-(N-methylmethylsulfonamido)-N3-((R)-1-phenylethyl)isophthalamide |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C46H59N5O8S |
|---|
| Mol. Mass. | 842.054 |
|---|
| SMILES | CC(C)[C@H](C[C@H](O)[C@H](COCc1ccccc1)NC(=O)c1cc(cc(c1)C(=O)N[C@H](C)c1ccccc1)N(C)S(C)(=O)=O)C(=O)N[C@@H](C(C)C)C(=O)NCc1ccccc1 |r| |
|---|
| Structure | 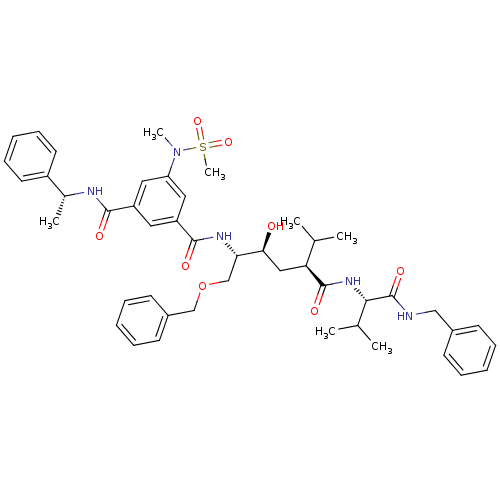
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Sund, C; Belda, O; Wiktelius, D; Sahlberg, C; Vrang, L; Sedig, S; Hamelink, E; Henderson, I; Agback, T; Jansson, K; Borkakoti, N; Derbyshire, D; Eneroth, A; Samuelsson, B Design and synthesis of potent macrocyclic renin inhibitors. Bioorg Med Chem Lett21:358-62 (2010) [PubMed] Article
Sund, C; Belda, O; Wiktelius, D; Sahlberg, C; Vrang, L; Sedig, S; Hamelink, E; Henderson, I; Agback, T; Jansson, K; Borkakoti, N; Derbyshire, D; Eneroth, A; Samuelsson, B Design and synthesis of potent macrocyclic renin inhibitors. Bioorg Med Chem Lett21:358-62 (2010) [PubMed] Article