

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
| Reaction Details | |||
|---|---|---|---|
 | Report a problem with these data | ||
| Target | Bile acid receptor | ||
| Ligand | BDBM50336388 | ||
| Substrate/Competitor | n/a | ||
| Meas. Tech. | ChEMBL_716784 (CHEMBL1670706) | ||
| IC50 | 210±n/a nM | ||
| Citation |  Richter, HG; Benson, GM; Bleicher, KH; Blum, D; Chaput, E; Clemann, N; Feng, S; Gardes, C; Grether, U; Hartman, P; Kuhn, B; Martin, RE; Plancher, JM; Rudolph, MG; Schuler, F; Taylor, S Optimization of a novel class of benzimidazole-based farnesoid X receptor (FXR) agonists to improve physicochemical and ADME properties. Bioorg Med Chem Lett21:1134-40 (2011) [PubMed] Article Richter, HG; Benson, GM; Bleicher, KH; Blum, D; Chaput, E; Clemann, N; Feng, S; Gardes, C; Grether, U; Hartman, P; Kuhn, B; Martin, RE; Plancher, JM; Rudolph, MG; Schuler, F; Taylor, S Optimization of a novel class of benzimidazole-based farnesoid X receptor (FXR) agonists to improve physicochemical and ADME properties. Bioorg Med Chem Lett21:1134-40 (2011) [PubMed] Article | ||
| More Info.: | Get all data from this article, Assay Method | ||
| Bile acid receptor | |||
| Name: | Bile acid receptor | ||
| Synonyms: | BAR | Bile acid receptor FXR | FXR | Farnesol receptor HRR-1 | HRR1 | NR1H4 | NR1H4_HUMAN | Nuclear receptor subfamily 1 group H member 4 | RIP14 | RXR-interacting protein 14 | Retinoid X receptor-interacting protein 14 | farnesoid x receptor | ||
| Type: | Nuclear Receptor | ||
| Mol. Mass.: | 55916.24 | ||
| Organism: | Homo sapiens (Human) | ||
| Description: | Q96RI1 | ||
| Residue: | 486 | ||
| Sequence: |
| ||
| BDBM50336388 | |||
| n/a | |||
| Name | BDBM50336388 | ||
| Synonyms: | CHEMBL1668261 | FXR_64 | trans-4-((2S)-2-cyclohexyl-2-(2-(2,4-dimethoxypyridin-3-yl)-5,6-difluoro-1H-benzo[d]imidazol-1-yl)acetamido)cyclohexanecarboxylic acid | ||
| Type | Small organic molecule | ||
| Emp. Form. | C29H34F2N4O5 | ||
| Mol. Mass. | 556.6009 | ||
| SMILES | COc1ccc(-c2nc3cc(F)c(F)cc3n2[C@@H](C2CCCCC2)C(=O)N[C@H]2CC[C@@H](CC2)C(O)=O)c(OC)n1 |r,wU:17.18,27.29,wD:30.36,(11.8,-19.09,;11.04,-17.76,;9.49,-17.74,;8.72,-19.08,;7.17,-19.07,;6.42,-17.75,;4.41,-17.75,;3.93,-16.28,;2.39,-16.28,;1.33,-15.13,;-.15,-15.46,;-1.19,-14.32,;-.62,-16.92,;-2.12,-17.24,;.41,-18.07,;1.92,-17.75,;3.17,-18.65,;3.17,-20.21,;4.5,-20.98,;4.5,-22.52,;5.84,-23.29,;7.17,-22.52,;7.17,-20.98,;5.84,-20.21,;1.83,-20.98,;1.83,-22.52,;.5,-20.21,;-.84,-20.98,;-.84,-22.53,;-2.16,-23.29,;-3.47,-22.54,;-3.47,-21,;-2.17,-20.21,;-4.83,-23.32,;-6.17,-22.55,;-4.83,-24.88,;7.19,-16.41,;6.42,-15.07,;7.12,-13.7,;8.73,-16.41,)| | ||
| Structure | 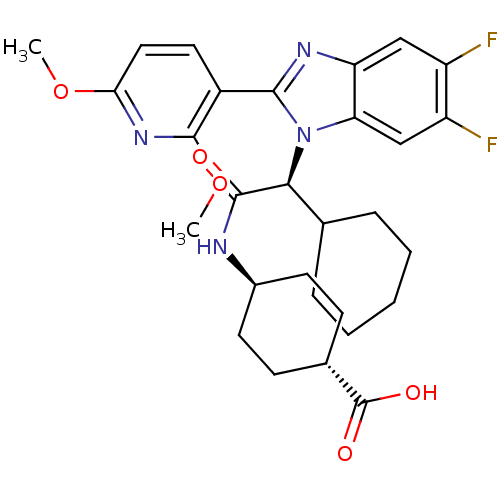
| ||