| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Prothrombin |
|---|
| Ligand | BDBM50337458 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_728384 (CHEMBL1685160) |
|---|
| Ki | 1.1±n/a nM |
|---|
| Citation |  Isaacs, RC; Newton, CL; Cutrona, KJ; Mercer, SP; Dorsey, BD; McDonough, CM; Cook, JJ; Krueger, JA; Lewis, SD; Lucas, BJ; Lyle, EA; Lynch, JJ; Miller-Stein, C; Michener, MT; Wallace, AA; White, RB; Wong, BK P3 optimization of functional potency, in vivo efficacy and oral bioavailability in 3-aminopyrazinone thrombin inhibitors bearing non-charged groups at the P1 position. Bioorg Med Chem Lett21:1532-5 (2011) [PubMed] Article Isaacs, RC; Newton, CL; Cutrona, KJ; Mercer, SP; Dorsey, BD; McDonough, CM; Cook, JJ; Krueger, JA; Lewis, SD; Lucas, BJ; Lyle, EA; Lynch, JJ; Miller-Stein, C; Michener, MT; Wallace, AA; White, RB; Wong, BK P3 optimization of functional potency, in vivo efficacy and oral bioavailability in 3-aminopyrazinone thrombin inhibitors bearing non-charged groups at the P1 position. Bioorg Med Chem Lett21:1532-5 (2011) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Prothrombin |
|---|
| Name: | Prothrombin |
|---|
| Synonyms: | Activation peptide fragment 1 | Activation peptide fragment 2 | Coagulation factor II | F2 | Prothrombin precursor | THRB_HUMAN | Thrombin heavy chain | Thrombin light chain |
|---|
| Type: | Protein |
|---|
| Mol. Mass.: | 70029.57 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | P00734 |
|---|
| Residue: | 622 |
|---|
| Sequence: | MAHVRGLQLPGCLALAALCSLVHSQHVFLAPQQARSLLQRVRRANTFLEEVRKGNLEREC
VEETCSYEEAFEALESSTATDVFWAKYTACETARTPRDKLAACLEGNCAEGLGTNYRGHV
NITRSGIECQLWRSRYPHKPEINSTTHPGADLQENFCRNPDSSTTGPWCYTTDPTVRRQE
CSIPVCGQDQVTVAMTPRSEGSSVNLSPPLEQCVPDRGQQYQGRLAVTTHGLPCLAWASA
QAKALSKHQDFNSAVQLVENFCRNPDGDEEGVWCYVAGKPGDFGYCDLNYCEEAVEEETG
DGLDEDSDRAIEGRTATSEYQTFFNPRTFGSGEADCGLRPLFEKKSLEDKTERELLESYI
DGRIVEGSDAEIGMSPWQVMLFRKSPQELLCGASLISDRWVLTAAHCLLYPPWDKNFTEN
DLLVRIGKHSRTRYERNIEKISMLEKIYIHPRYNWRENLDRDIALMKLKKPVAFSDYIHP
VCLPDRETAASLLQAGYKGRVTGWGNLKETWTANVGKGQPSVLQVVNLPIVERPVCKDST
RIRITDNMFCAGYKPDEGKRGDACEGDSGGPFVMKSPFNNRWYQMGIVSWGEGCDRDGKY
GFYTHVFRLKKWIQKVIDQFGE
|
|
|
|---|
| BDBM50337458 |
|---|
| n/a |
|---|
| Name | BDBM50337458 |
|---|
| Synonyms: | CHEMBL1682697 | N-(5-chloro-2-methylbenzyl)-2-(6-methyl-2-oxo-3-(2-(pyridin-2-yl)ethylamino)pyrazin-1(2H)-yl)acetamide |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C22H24ClN5O2 |
|---|
| Mol. Mass. | 425.911 |
|---|
| SMILES | Cc1ccc(Cl)cc1CNC(=O)Cn1c(C)cnc(NCCc2ccccn2)c1=O |
|---|
| Structure | 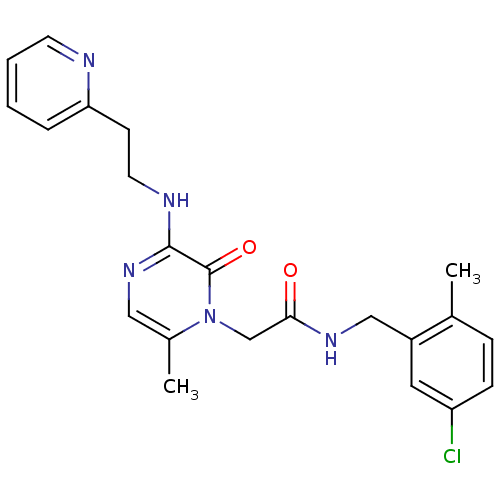
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Isaacs, RC; Newton, CL; Cutrona, KJ; Mercer, SP; Dorsey, BD; McDonough, CM; Cook, JJ; Krueger, JA; Lewis, SD; Lucas, BJ; Lyle, EA; Lynch, JJ; Miller-Stein, C; Michener, MT; Wallace, AA; White, RB; Wong, BK P3 optimization of functional potency, in vivo efficacy and oral bioavailability in 3-aminopyrazinone thrombin inhibitors bearing non-charged groups at the P1 position. Bioorg Med Chem Lett21:1532-5 (2011) [PubMed] Article
Isaacs, RC; Newton, CL; Cutrona, KJ; Mercer, SP; Dorsey, BD; McDonough, CM; Cook, JJ; Krueger, JA; Lewis, SD; Lucas, BJ; Lyle, EA; Lynch, JJ; Miller-Stein, C; Michener, MT; Wallace, AA; White, RB; Wong, BK P3 optimization of functional potency, in vivo efficacy and oral bioavailability in 3-aminopyrazinone thrombin inhibitors bearing non-charged groups at the P1 position. Bioorg Med Chem Lett21:1532-5 (2011) [PubMed] Article