| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Progesterone receptor |
|---|
| Ligand | BDBM50338693 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_726893 (CHEMBL1686599) |
|---|
| IC50 | 130±n/a nM |
|---|
| Citation |  Nagata, N; Miyakawa, M; Amano, S; Furuya, K; Yamamoto, N; Inoguchi, K Design and synthesis of tricyclic tetrahydroquinolines as a new series of nonsteroidal selective androgen receptor modulators (SARMs). Bioorg Med Chem Lett21:1744-7 (2011) [PubMed] Article Nagata, N; Miyakawa, M; Amano, S; Furuya, K; Yamamoto, N; Inoguchi, K Design and synthesis of tricyclic tetrahydroquinolines as a new series of nonsteroidal selective androgen receptor modulators (SARMs). Bioorg Med Chem Lett21:1744-7 (2011) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Progesterone receptor |
|---|
| Name: | Progesterone receptor |
|---|
| Synonyms: | NR3C3 | PGR | PRGR_RABIT |
|---|
| Type: | PROTEIN |
|---|
| Mol. Mass.: | 98665.51 |
|---|
| Organism: | Oryctolagus cuniculus |
|---|
| Description: | ChEMBL_775980 |
|---|
| Residue: | 930 |
|---|
| Sequence: | MTELKAKEPRAPHVAGGAPSPTEVGSQLLGRPDPGPFQGSQTSEASSVVSAIPISLDGLL
FPRPCQGQNPPDGKTQDPPSLSDVEGAFPGVEAPEGAGDSSSRPPEKDSGLLDSVLDTLL
APSGPGQSHASPATCEAISPWCLFGPDLPEDPRAAPATKGVLAPLMSRPEDKAGDSSGTA
AAHKVLPRGLSPSRQLLLPSSGSPHWPAVKPSPQPAAVQVDEEDSSESEGTVGPLLKGQP
RALGGTAAGGGAAPVASGAAAGGVALVPKEDSRFSAPRVSLAEQDAPVAPGRSPLATSVV
DFIHVPILPLNHAFLATRTRQLLEGESYDGGAAAASPFVPQRGSPSASSTPVAGGDFPDC
TYPPDAEPKDDAFPLYGDFQPPALKIKEEEEAAEAAARSPRTYLVAGANPAAFPDFQLAA
PPPPSLPPRVPSSRPGEAAVAASPGSASVSSSSSSGSTLECILYKAEGAPPQQGPFAPLP
CKPPGAGACLLPRDGLPSTSASGAAAGAAPALYPTLGLNGLPQLGYQAAVLKEGLPQVYT
PYLNYLRPDSEASQSPQYSFESLPQKICLICGDEASGCHYGVLTCGSCKVFFKRAMEGQH
NYLCAGRNDCIVDKIRRKNCPACRLRKCCQAGMVLGGRKFKKFNKVRVMRALDAVALPQP
VGIPNESQRITFSPSQEIQLIPPLINLLMSIEPDVIYAGHDNTKPDTSSSLLTSLNQLGE
RQLLSVVKWSKSLPGFRNLHIDDQITLIQYSWMSLMVFGLGWRSYKHVSGQMLYFAPDLI
LNEQRMKESSFYSLCLTMWQIPQEFVKLQVSQEEFLCMKVLLLLNTIPLEGLRSQSQFEE
MRSSYIRELIKAIGLRQKGVVSSSQRFYQLTKLLDNLHDLVKQLHLYCLNTFIQSRALSV
EFPEMMSEVIAAQLPKILAGMVKPLLFHKK
|
|
|
|---|
| BDBM50338693 |
|---|
| n/a |
|---|
| Name | BDBM50338693 |
|---|
| Synonyms: | 2-methyl-2-((3aR,4S,9bS)-8-nitro-3a,4,5,9b-tetrahydro-3H-cyclopenta[c]quinolin-4-yl)propan-1-ol | CHEMBL1684194 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C16H20N2O3 |
|---|
| Mol. Mass. | 288.3416 |
|---|
| SMILES | CC(C)(CO)[C@H]1Nc2ccc(cc2[C@H]2C=CC[C@@H]12)[N+]([O-])=O |r,c:15| |
|---|
| Structure | 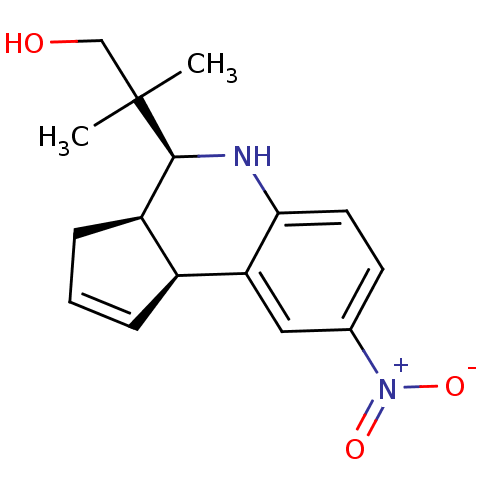
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Nagata, N; Miyakawa, M; Amano, S; Furuya, K; Yamamoto, N; Inoguchi, K Design and synthesis of tricyclic tetrahydroquinolines as a new series of nonsteroidal selective androgen receptor modulators (SARMs). Bioorg Med Chem Lett21:1744-7 (2011) [PubMed] Article
Nagata, N; Miyakawa, M; Amano, S; Furuya, K; Yamamoto, N; Inoguchi, K Design and synthesis of tricyclic tetrahydroquinolines as a new series of nonsteroidal selective androgen receptor modulators (SARMs). Bioorg Med Chem Lett21:1744-7 (2011) [PubMed] Article