| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Carbonic anhydrase 9 |
|---|
| Ligand | BDBM54376 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_735541 (CHEMBL1693046) |
|---|
| Ki | 58±n/a nM |
|---|
| Citation |  Pacchiano, F; Carta, F; McDonald, PC; Lou, Y; Vullo, D; Scozzafava, A; Dedhar, S; Supuran, CT Ureido-substituted benzenesulfonamides potently inhibit carbonic anhydrase IX and show antimetastatic activity in a model of breast cancer metastasis. J Med Chem54:1896-902 (2011) [PubMed] Article Pacchiano, F; Carta, F; McDonald, PC; Lou, Y; Vullo, D; Scozzafava, A; Dedhar, S; Supuran, CT Ureido-substituted benzenesulfonamides potently inhibit carbonic anhydrase IX and show antimetastatic activity in a model of breast cancer metastasis. J Med Chem54:1896-902 (2011) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Carbonic anhydrase 9 |
|---|
| Name: | Carbonic anhydrase 9 |
|---|
| Synonyms: | CA-IX | CA9 | CAH9_HUMAN | Carbonate dehydratase IX | Carbonic anhydrase 9 (CA IX) | Carbonic anhydrase 9 (CAIX) | Carbonic anhydrase 9 precursor | Carbonic anhydrase IX (CA IX) | Carbonic anhydrase IX (CAIX) | Carbonic anhydrases IX | Carbonic anhydrases; II & IX | G250 | MN | Membrane antigen MN | RCC-associated antigen G250 |
|---|
| Type: | Enzyme |
|---|
| Mol. Mass.: | 49669.03 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | Catalytic domain of human cloned isozyme was used in the assay |
|---|
| Residue: | 459 |
|---|
| Sequence: | MAPLCPSPWLPLLIPAPAPGLTVQLLLSLLLLVPVHPQRLPRMQEDSPLGGGSSGEDDPL
GEEDLPSEEDSPREEDPPGEEDLPGEEDLPGEEDLPEVKPKSEEEGSLKLEDLPTVEAPG
DPQEPQNNAHRDKEGDDQSHWRYGGDPPWPRVSPACAGRFQSPVDIRPQLAAFCPALRPL
ELLGFQLPPLPELRLRNNGHSVQLTLPPGLEMALGPGREYRALQLHLHWGAAGRPGSEHT
VEGHRFPAEIHVVHLSTAFARVDEALGRPGGLAVLAAFLEEGPEENSAYEQLLSRLEEIA
EEGSETQVPGLDISALLPSDFSRYFQYEGSLTTPPCAQGVIWTVFNQTVMLSAKQLHTLS
DTLWGPGDSRLQLNFRATQPLNGRVIEASFPAGVDSSPRAAEPVQLNSCLAAGDILALVF
GLLFAVTSVAFLVQMRRQHRRGTKGGVSYRPAEVAETGA
|
|
|
|---|
| BDBM54376 |
|---|
| n/a |
|---|
| Name | BDBM54376 |
|---|
| Synonyms: | 6-Hexyl-7-hydroxy-2-oxo-2H-chromene-3-carboxylic acid ethyl ester | 6-hexyl-7-hydroxy-2-keto-chromene-3-carboxylic acid ethyl ester | 6-hexyl-7-hydroxy-2-oxo-1-benzopyran-3-carboxylic acid ethyl ester | MLS000777587 | SMR000413981 | cid_5545047 | ethyl 6-hexyl-7-hydroxy-2-oxochromene-3-carboxylate | ethyl 6-hexyl-7-oxidanyl-2-oxidanylidene-chromene-3-carboxylate |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C18H22O5 |
|---|
| Mol. Mass. | 318.3643 |
|---|
| SMILES | CCCCCCc1cc2cc(C(=O)OCC)c(=O)oc2cc1O |
|---|
| Structure | 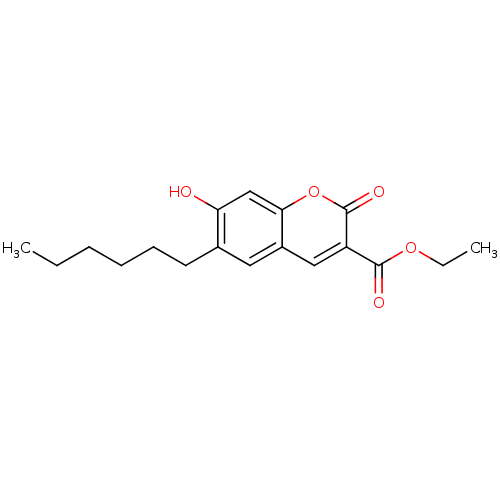
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Pacchiano, F; Carta, F; McDonald, PC; Lou, Y; Vullo, D; Scozzafava, A; Dedhar, S; Supuran, CT Ureido-substituted benzenesulfonamides potently inhibit carbonic anhydrase IX and show antimetastatic activity in a model of breast cancer metastasis. J Med Chem54:1896-902 (2011) [PubMed] Article
Pacchiano, F; Carta, F; McDonald, PC; Lou, Y; Vullo, D; Scozzafava, A; Dedhar, S; Supuran, CT Ureido-substituted benzenesulfonamides potently inhibit carbonic anhydrase IX and show antimetastatic activity in a model of breast cancer metastasis. J Med Chem54:1896-902 (2011) [PubMed] Article