| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Cannabinoid receptor 1 |
|---|
| Ligand | BDBM50341920 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_743423 (CHEMBL1767777) |
|---|
| IC50 | 930±n/a nM |
|---|
| Citation |  Manley, PJ; Zartman, A; Paone, DV; Burgey, CS; Henze, DA; Della Penna, K; Desai, R; Leitl, MD; Lemaire, W; White, RB; Yeh, S; Urban, MO; Kane, SA; Hartman, GD; Bilodeau, MT; Trotter, BW Decahydroquinoline amides as highly selective CB2 agonists: role of selectivity on in vivo efficacy in a rodent model of analgesia. Bioorg Med Chem Lett21:2359-64 (2011) [PubMed] Article Manley, PJ; Zartman, A; Paone, DV; Burgey, CS; Henze, DA; Della Penna, K; Desai, R; Leitl, MD; Lemaire, W; White, RB; Yeh, S; Urban, MO; Kane, SA; Hartman, GD; Bilodeau, MT; Trotter, BW Decahydroquinoline amides as highly selective CB2 agonists: role of selectivity on in vivo efficacy in a rodent model of analgesia. Bioorg Med Chem Lett21:2359-64 (2011) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Cannabinoid receptor 1 |
|---|
| Name: | Cannabinoid receptor 1 |
|---|
| Synonyms: | CANN6 | CANNABINOID CB1 | CB-R | CB1 | CNR | CNR1 | CNR1_HUMAN | Cannabinoid CB1 receptor | Cannabinoid receptor | Cannabinoid receptor 1 (CB1) | Cannabinoid receptor 1 (brain) |
|---|
| Type: | G Protein-Coupled Receptor (GPCR) |
|---|
| Mol. Mass.: | 52868.96 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | P21554 |
|---|
| Residue: | 472 |
|---|
| Sequence: | MKSILDGLADTTFRTITTDLLYVGSNDIQYEDIKGDMASKLGYFPQKFPLTSFRGSPFQE
KMTAGDNPQLVPADQVNITEFYNKSLSSFKENEENIQCGENFMDIECFMVLNPSQQLAIA
VLSLTLGTFTVLENLLVLCVILHSRSLRCRPSYHFIGSLAVADLLGSVIFVYSFIDFHVF
HRKDSRNVFLFKLGGVTASFTASVGSLFLTAIDRYISIHRPLAYKRIVTRPKAVVAFCLM
WTIAIVIAVLPLLGWNCEKLQSVCSDIFPHIDETYLMFWIGVTSVLLLFIVYAYMYILWK
AHSHAVRMIQRGTQKSIIIHTSEDGKVQVTRPDQARMDIRLAKTLVLILVVLIICWGPLL
AIMVYDVFGKMNKLIKTVFAFCSMLCLLNSTVNPIIYALRSKDLRHAFRSMFPSCEGTAQ
PLDNSMGDSDCLHKHANNAASVHRAAESCIKSTVKIAKVTMSVSTDTSAEAL
|
|
|
|---|
| BDBM50341920 |
|---|
| n/a |
|---|
| Name | BDBM50341920 |
|---|
| Synonyms: | ((4aR,8aS)-4-hydroxy-4-phenyloctahydroquinolin-1(2H)-yl)(2-(pyridin-4-yl)thiazol-4-yl)methanone | ((4aR,8aS)-4-hydroxy-4-phenyloctahydroquinolin-1(2H)-yl)(pyridin-2-yl)methanone | (3-(2-fluorophenyl)-1H-pyrazol-5-yl)((4aR,8aS)-4-hydroxy-4-phenyloctahydroquinolin-1(2H)-yl)methanone | CHEMBL1765092 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C24H25N3O2S |
|---|
| Mol. Mass. | 419.539 |
|---|
| SMILES | OC1(CCN([C@H]2CCCC[C@@H]12)C(=O)c1csc(n1)-c1ccncc1)c1ccccc1 |r| |
|---|
| Structure | 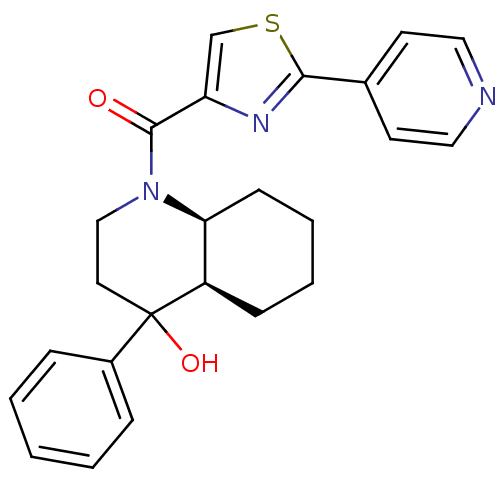
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Manley, PJ; Zartman, A; Paone, DV; Burgey, CS; Henze, DA; Della Penna, K; Desai, R; Leitl, MD; Lemaire, W; White, RB; Yeh, S; Urban, MO; Kane, SA; Hartman, GD; Bilodeau, MT; Trotter, BW Decahydroquinoline amides as highly selective CB2 agonists: role of selectivity on in vivo efficacy in a rodent model of analgesia. Bioorg Med Chem Lett21:2359-64 (2011) [PubMed] Article
Manley, PJ; Zartman, A; Paone, DV; Burgey, CS; Henze, DA; Della Penna, K; Desai, R; Leitl, MD; Lemaire, W; White, RB; Yeh, S; Urban, MO; Kane, SA; Hartman, GD; Bilodeau, MT; Trotter, BW Decahydroquinoline amides as highly selective CB2 agonists: role of selectivity on in vivo efficacy in a rodent model of analgesia. Bioorg Med Chem Lett21:2359-64 (2011) [PubMed] Article