| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Phosphatidylinositol 3-kinase catalytic subunit type 3 |
|---|
| Ligand | BDBM50343200 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_745554 (CHEMBL1775871) |
|---|
| Ki | 31±n/a nM |
|---|
| Citation |  D'Angelo, ND; Kim, TS; Andrews, K; Booker, SK; Caenepeel, S; Chen, K; D'Amico, D; Freeman, D; Jiang, J; Liu, L; McCarter, JD; San Miguel, T; Mullady, EL; Schrag, M; Subramanian, R; Tang, J; Wahl, RC; Wang, L; Whittington, DA; Wu, T; Xi, N; Xu, Y; Yakowec, P; Yang, K; Zalameda, LP; Zhang, N; Hughes, P; Norman, MH Discovery and optimization of a series of benzothiazole phosphoinositide 3-kinase (PI3K)/mammalian target of rapamycin (mTOR) dual inhibitors. J Med Chem54:1789-811 (2011) [PubMed] Article D'Angelo, ND; Kim, TS; Andrews, K; Booker, SK; Caenepeel, S; Chen, K; D'Amico, D; Freeman, D; Jiang, J; Liu, L; McCarter, JD; San Miguel, T; Mullady, EL; Schrag, M; Subramanian, R; Tang, J; Wahl, RC; Wang, L; Whittington, DA; Wu, T; Xi, N; Xu, Y; Yakowec, P; Yang, K; Zalameda, LP; Zhang, N; Hughes, P; Norman, MH Discovery and optimization of a series of benzothiazole phosphoinositide 3-kinase (PI3K)/mammalian target of rapamycin (mTOR) dual inhibitors. J Med Chem54:1789-811 (2011) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Phosphatidylinositol 3-kinase catalytic subunit type 3 |
|---|
| Name: | Phosphatidylinositol 3-kinase catalytic subunit type 3 |
|---|
| Synonyms: | BC033004 | PI3K type 3 | PIK3C3 | PK3C3_HUMAN | Phosphatidylinositol 3-kinase catalytic subunit type 3 (PIK3C3) | Phosphatidylinositol 3-kinase p100 subunit | Phosphoinositide 3-Kinase (PI3K), Vps34 | Phosphoinositide-3-kinase class 3 | PtdIns-3-kinase type 3 | VPS34 | hVPS34 |
|---|
| Type: | Enzyme |
|---|
| Mol. Mass.: | 101551.30 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | Recombinant human Vps34 protein was expressed as monomeric GST-fusion and purified. |
|---|
| Residue: | 887 |
|---|
| Sequence: | MGEAEKFHYIYSCDLDINVQLKIGSLEGKREQKSYKAVLEDPMLKFSGLYQETCSDLYVT
CQVFAEGKPLALPVRTSYKAFSTRWNWNEWLKLPVKYPDLPRNAQVALTIWDVYGPGKAV
PVGGTTVSLFGKYGMFRQGMHDLKVWPNVEADGSEPTKTPGRTSSTLSEDQMSRLAKLTK
AHRQGHMVKVDWLDRLTFREIEMINESEKRSSNFMYLMVEFRCVKCDDKEYGIVYYEKDG
DESSPILTSFELVKVPDPQMSMENLVESKHHKLARSLRSGPSDHDLKPNAATRDQLNIIV
SYPPTKQLTYEEQDLVWKFRYYLTNQEKALTKFLKCVNWDLPQEAKQALELLGKWKPMDV
EDSLELLSSHYTNPTVRRYAVARLRQADDEDLLMYLLQLVQALKYENFDDIKNGLEPTKK
DSQSSVSENVSNSGINSAEIDSSQIITSPLPSVSSPPPASKTKEVPDGENLEQDLCTFLI
SRACKNSTLANYLYWYVIVECEDQDTQQRDPKTHEMYLNVMRRFSQALLKGDKSVRVMRS
LLAAQQTFVDRLVHLMKAVQRESGNRKKKNERLQALLGDNEKMNLSDVELIPLPLEPQVK
IRGIIPETATLFKSALMPAQLFFKTEDGGKYPVIFKHGDDLRQDQLILQIISLMDKLLRK
ENLDLKLTPYKVLATSTKHGFMQFIQSVPVAEVLDTEGSIQNFFRKYAPSENGPNGISAE
VMDTYVKSCAGYCVITYILGVGDRHLDNLLLTKTGKLFHIDFGYILGRDPKPLPPPMKLN
KEMVEGMGGTQSEQYQEFRKQCYTAFLHLRRYSNLILNLFSLMVDANIPDIALEPDKTVK
KVQDKFRLDLSDEEAVHYMQSLIDESVHALFAAVVEQIHKFAQYWRK
|
|
|
|---|
| BDBM50343200 |
|---|
| n/a |
|---|
| Name | BDBM50343200 |
|---|
| Synonyms: | CHEMBL1615189 | N-(6-(6-Chloro-5-(4-fluorophenylsulfonamido)pyridin-3-yl)benzo[d]thiazol-2-yl)acetamide | N-[6-(6-chloro-5-{[(4-fluorophenyl)sulfonyl]amino}pyridin-3-yl)-1,3-benzothiazol-2-yl]acetamide |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C20H14ClFN4O3S2 |
|---|
| Mol. Mass. | 476.932 |
|---|
| SMILES | CC(=O)Nc1nc2ccc(cc2s1)-c1cnc(Cl)c(NS(=O)(=O)c2ccc(F)cc2)c1 |
|---|
| Structure | 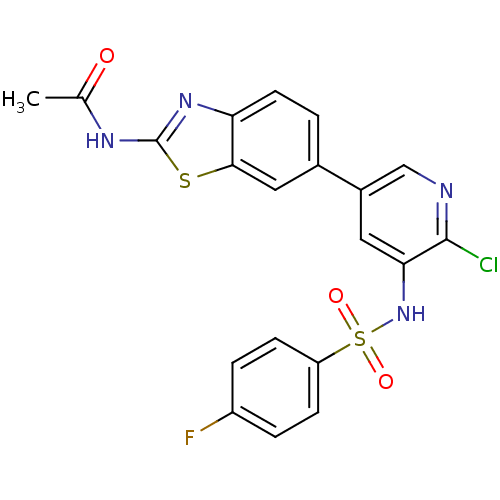
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 D'Angelo, ND; Kim, TS; Andrews, K; Booker, SK; Caenepeel, S; Chen, K; D'Amico, D; Freeman, D; Jiang, J; Liu, L; McCarter, JD; San Miguel, T; Mullady, EL; Schrag, M; Subramanian, R; Tang, J; Wahl, RC; Wang, L; Whittington, DA; Wu, T; Xi, N; Xu, Y; Yakowec, P; Yang, K; Zalameda, LP; Zhang, N; Hughes, P; Norman, MH Discovery and optimization of a series of benzothiazole phosphoinositide 3-kinase (PI3K)/mammalian target of rapamycin (mTOR) dual inhibitors. J Med Chem54:1789-811 (2011) [PubMed] Article
D'Angelo, ND; Kim, TS; Andrews, K; Booker, SK; Caenepeel, S; Chen, K; D'Amico, D; Freeman, D; Jiang, J; Liu, L; McCarter, JD; San Miguel, T; Mullady, EL; Schrag, M; Subramanian, R; Tang, J; Wahl, RC; Wang, L; Whittington, DA; Wu, T; Xi, N; Xu, Y; Yakowec, P; Yang, K; Zalameda, LP; Zhang, N; Hughes, P; Norman, MH Discovery and optimization of a series of benzothiazole phosphoinositide 3-kinase (PI3K)/mammalian target of rapamycin (mTOR) dual inhibitors. J Med Chem54:1789-811 (2011) [PubMed] Article