| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Cholinesterase |
|---|
| Ligand | BDBM13551 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_751135 (CHEMBL1787217) |
|---|
| IC50 | 840±n/a nM |
|---|
| Citation |  Tasso, B; Catto, M; Nicolotti, O; Novelli, F; Tonelli, M; Giangreco, I; Pisani, L; Sparatore, A; Boido, V; Carotti, A; Sparatore, F Quinolizidinyl derivatives of bi- and tricyclic systems as potent inhibitors of acetyl- and butyrylcholinesterase with potential in Alzheimer's disease. Eur J Med Chem46:2170-84 (2011) [PubMed] Article Tasso, B; Catto, M; Nicolotti, O; Novelli, F; Tonelli, M; Giangreco, I; Pisani, L; Sparatore, A; Boido, V; Carotti, A; Sparatore, F Quinolizidinyl derivatives of bi- and tricyclic systems as potent inhibitors of acetyl- and butyrylcholinesterase with potential in Alzheimer's disease. Eur J Med Chem46:2170-84 (2011) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Cholinesterase |
|---|
| Name: | Cholinesterase |
|---|
| Synonyms: | BCHE | Butyrylcholinesterase (BuChE) | CHLE_HORSE | Cholinesterase |
|---|
| Type: | Enzyme |
|---|
| Mol. Mass.: | 65643.35 |
|---|
| Organism: | Equus caballus (Horse) |
|---|
| Description: | P81908 |
|---|
| Residue: | 574 |
|---|
| Sequence: | EEDIIITTKNGKVRGMNLPVLGGTVTAFLGIPYAQPPLGRLRFKKPQSLTKWSNIWNATK
YANSCYQNTDQSFPGFLGSEMWNPNTELSEDCLYLNVWIPAPKPKNATVMIWIYGGGFQT
GTSSLPVYDGKFLARVERVIVVSMNYRVGALGFLALSENPEAPGNMGLFDQQLALQWVQK
NIAAFGGNPRSVTLFGESAGAASVSLHLLSPRSQPLFTRAILQSGSSNAPWAVTSLYEAR
NRTLTLAKRMGCSRDNETEMIKCLRDKDPQEILLNEVFVVPYDTLLSVNFGPTVDGDFLT
DMPDTLLQLGQFKRTQILVGVNKDEGTAFLVYGAPGFSKDNNSIITRKEFQEGLKIFFPR
VSEFGRESILFHYMDWLDDQRAENYREALDDVVGDYNIICPALEFTRKFSELGNDAFFYY
FEHRSTKLPWPEWMGVMHGYEIEFVFGLPLERRVNYTRAEEILSRSIMKRWANFAKYGNP
NGTQNNSTRWPVFKSTEQKYLTLNTESPKVYTKLRAQQCRFWTLFFPKVLELTGNIDEAE
REWKAGFHRWNNYMMDWKNQFNDYTSKKESCSDF
|
|
|
|---|
| BDBM13551 |
|---|
| n/a |
|---|
| Name | BDBM13551 |
|---|
| Synonyms: | 10-[2-(Diethylamino)-1-oxopropyl]-10H-phenothiazine | 2-(diethylamino)-1-(10H-phenothiazin-10-yl)propan-1-one | AS-1397 | CHEMBL1782730 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C19H22N2OS |
|---|
| Mol. Mass. | 326.456 |
|---|
| SMILES | CCN(CC)C(C)C(=O)N1c2ccccc2Sc2ccccc12 |
|---|
| Structure | 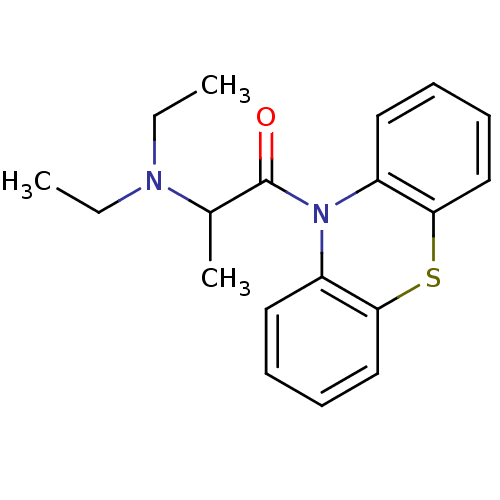
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Tasso, B; Catto, M; Nicolotti, O; Novelli, F; Tonelli, M; Giangreco, I; Pisani, L; Sparatore, A; Boido, V; Carotti, A; Sparatore, F Quinolizidinyl derivatives of bi- and tricyclic systems as potent inhibitors of acetyl- and butyrylcholinesterase with potential in Alzheimer's disease. Eur J Med Chem46:2170-84 (2011) [PubMed] Article
Tasso, B; Catto, M; Nicolotti, O; Novelli, F; Tonelli, M; Giangreco, I; Pisani, L; Sparatore, A; Boido, V; Carotti, A; Sparatore, F Quinolizidinyl derivatives of bi- and tricyclic systems as potent inhibitors of acetyl- and butyrylcholinesterase with potential in Alzheimer's disease. Eur J Med Chem46:2170-84 (2011) [PubMed] Article