| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Cytochrome P450 3A4 |
|---|
| Ligand | BDBM50353232 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_769156 (CHEMBL1832815) |
|---|
| IC50 | 330±n/a nM |
|---|
| Citation |  Wang, H; Yu, N; Chen, D; Lee, KC; Lye, PL; Chang, JW; Deng, W; Ng, MC; Lu, T; Khoo, ML; Poulsen, A; Sangthongpitag, K; Wu, X; Hu, C; Goh, KC; Wang, X; Fang, L; Goh, KL; Khng, HH; Goh, SK; Yeo, P; Liu, X; Bonday, Z; Wood, JM; Dymock, BW; Kantharaj, E; Sun, ET Discovery of (2E)-3-{2-butyl-1-[2-(diethylamino)ethyl]-1H-benzimidazol-5-yl}-N-hydroxyacrylamide (SB939), an orally active histone deacetylase inhibitor with a superior preclinical profile. J Med Chem54:4694-720 (2011) [PubMed] Article Wang, H; Yu, N; Chen, D; Lee, KC; Lye, PL; Chang, JW; Deng, W; Ng, MC; Lu, T; Khoo, ML; Poulsen, A; Sangthongpitag, K; Wu, X; Hu, C; Goh, KC; Wang, X; Fang, L; Goh, KL; Khng, HH; Goh, SK; Yeo, P; Liu, X; Bonday, Z; Wood, JM; Dymock, BW; Kantharaj, E; Sun, ET Discovery of (2E)-3-{2-butyl-1-[2-(diethylamino)ethyl]-1H-benzimidazol-5-yl}-N-hydroxyacrylamide (SB939), an orally active histone deacetylase inhibitor with a superior preclinical profile. J Med Chem54:4694-720 (2011) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Cytochrome P450 3A4 |
|---|
| Name: | Cytochrome P450 3A4 |
|---|
| Synonyms: | Albendazole monooxygenase | Albendazole sulfoxidase | CP3A4_HUMAN | CYP3A3 | CYP3A4 | CYPIIIA3 | CYPIIIA4 | Cytochrome P450 3A3 | Cytochrome P450 3A4 (CYP3A4) | Cytochrome P450 HLp | Nifedipine oxidase | Quinine 3-monooxygenase | Taurochenodeoxycholate 6-alpha-hydroxylase |
|---|
| Type: | Enzyme |
|---|
| Mol. Mass.: | 57349.57 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | n/a |
|---|
| Residue: | 503 |
|---|
| Sequence: | MALIPDLAMETWLLLAVSLVLLYLYGTHSHGLFKKLGIPGPTPLPFLGNILSYHKGFCMF
DMECHKKYGKVWGFYDGQQPVLAITDPDMIKTVLVKECYSVFTNRRPFGPVGFMKSAISI
AEDEEWKRLRSLLSPTFTSGKLKEMVPIIAQYGDVLVRNLRREAETGKPVTLKDVFGAYS
MDVITSTSFGVNIDSLNNPQDPFVENTKKLLRFDFLDPFFLSITVFPFLIPILEVLNICV
FPREVTNFLRKSVKRMKESRLEDTQKHRVDFLQLMIDSQNSKETESHKALSDLELVAQSI
IFIFAGYETTSSVLSFIMYELATHPDVQQKLQEEIDAVLPNKAPPTYDTVLQMEYLDMVV
NETLRLFPIAMRLERVCKKDVEINGMFIPKGVVVMIPSYALHRDPKYWTEPEKFLPERFS
KKNKDNIDPYIYTPFGSGPRNCIGMRFALMNMKLALIRVLQNFSFKPCKETQIPLKLSLG
GLLQPEKPVVLKVESRDGTVSGA
|
|
|
|---|
| BDBM50353232 |
|---|
| n/a |
|---|
| Name | BDBM50353232 |
|---|
| Synonyms: | CHEMBL1830424 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C20H30N4O2 |
|---|
| Mol. Mass. | 358.4778 |
|---|
| SMILES | CCCCCCc1nc2cc(\C=C\C(=O)NO)ccc2n1CCN(C)C |
|---|
| Structure | 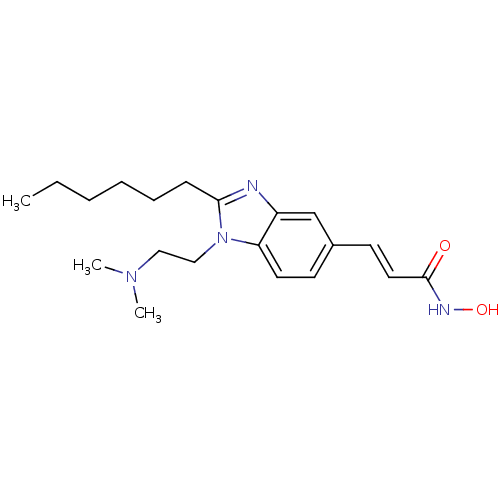
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Wang, H; Yu, N; Chen, D; Lee, KC; Lye, PL; Chang, JW; Deng, W; Ng, MC; Lu, T; Khoo, ML; Poulsen, A; Sangthongpitag, K; Wu, X; Hu, C; Goh, KC; Wang, X; Fang, L; Goh, KL; Khng, HH; Goh, SK; Yeo, P; Liu, X; Bonday, Z; Wood, JM; Dymock, BW; Kantharaj, E; Sun, ET Discovery of (2E)-3-{2-butyl-1-[2-(diethylamino)ethyl]-1H-benzimidazol-5-yl}-N-hydroxyacrylamide (SB939), an orally active histone deacetylase inhibitor with a superior preclinical profile. J Med Chem54:4694-720 (2011) [PubMed] Article
Wang, H; Yu, N; Chen, D; Lee, KC; Lye, PL; Chang, JW; Deng, W; Ng, MC; Lu, T; Khoo, ML; Poulsen, A; Sangthongpitag, K; Wu, X; Hu, C; Goh, KC; Wang, X; Fang, L; Goh, KL; Khng, HH; Goh, SK; Yeo, P; Liu, X; Bonday, Z; Wood, JM; Dymock, BW; Kantharaj, E; Sun, ET Discovery of (2E)-3-{2-butyl-1-[2-(diethylamino)ethyl]-1H-benzimidazol-5-yl}-N-hydroxyacrylamide (SB939), an orally active histone deacetylase inhibitor with a superior preclinical profile. J Med Chem54:4694-720 (2011) [PubMed] Article