| Reaction Details |
|---|
 | Report a problem with these data |
| Target | D(1B) dopamine receptor |
|---|
| Ligand | BDBM50004923 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_769762 (CHEMBL1831568) |
|---|
| Ki | 124±n/a nM |
|---|
| Citation |  Lebar, MD; Hahn, KN; Mutka, T; Maignan, P; McClintock, JB; Amsler, CD; van Olphen, A; Kyle, DE; Baker, BJ CNS and antimalarial activity of synthetic meridianin and psammopemmin analogs. Bioorg Med Chem19:5756-62 (2011) [PubMed] Article Lebar, MD; Hahn, KN; Mutka, T; Maignan, P; McClintock, JB; Amsler, CD; van Olphen, A; Kyle, DE; Baker, BJ CNS and antimalarial activity of synthetic meridianin and psammopemmin analogs. Bioorg Med Chem19:5756-62 (2011) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| D(1B) dopamine receptor |
|---|
| Name: | D(1B) dopamine receptor |
|---|
| Synonyms: | D(5) dopamine receptor | D1beta dopamine receptor | DOPAMINE D5 | DRD1B | DRD1L2 | DRD5 | DRD5_HUMAN | dopamine receptor D5 |
|---|
| Type: | Protein |
|---|
| Mol. Mass.: | 52943.41 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | P21918 |
|---|
| Residue: | 477 |
|---|
| Sequence: | MLPPGSNGTAYPGQFALYQQLAQGNAVGGSAGAPPLGPSQVVTACLLTLLIIWTLLGNVL
VCAAIVRSRHLRANMTNVFIVSLAVSDLFVALLVMPWKAVAEVAGYWPFGAFCDVWVAFD
IMCSTASILNLCVISVDRYWAISRPFRYKRKMTQRMALVMVGLAWTLSILISFIPVQLNW
HRDQAASWGGLDLPNNLANWTPWEEDFWEPDVNAENCDSSLNRTYAISSSLISFYIPVAI
MIVTYTRIYRIAQVQIRRISSLERAAEHAQSCRSSAACAPDTSLRASIKKETKVLKTLSV
IMGVFVCCWLPFFILNCMVPFCSGHPEGPPAGFPCVSETTFDVFVWFGWANSSLNPVIYA
FNADFQKVFAQLLGCSHFCSRTPVETVNISNELISYNQDIVFHKEIAAAYIHMMPNAVTP
GNREVDNDEEEGPFDRMFQIYQTSPDGDPVAESVWELDCEGEISLDKITPFTPNGFH
|
|
|
|---|
| BDBM50004923 |
|---|
| n/a |
|---|
| Name | BDBM50004923 |
|---|
| Synonyms: | (+/-)-SKF-38393 | 1-Phenyl-2,3,4,5-tetrahydro-1H-benzo[d]azepine-7,8-diol | 1-Phenyl-2,3,4,5-tetrahydro-1H-benzo[d]azepine-7,8-diol (SK&F 38393) | 1-Phenyl-2,3,4,5-tetrahydro-1H-benzo[d]azepine-7,8-diol(SKF 38393) | 1-Phenyl-2,3,4,5-tetrahydro-1H-benzo[d]azepine-7,8-diol; hydrochloride(SKF 38393) | CHEMBL286080 | CHEMBL505308 | CHEMBL542700 | RS(+/-)SKF 383931-Phenyl-2,3,4,5-tetrahydro-1H-benzo[d]azepine-7,8-diol | SK&F-38393 | SK-38393 | SKF 38393 | SKF-38393 | US9359372, SKF38393 | cid_147514 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C16H17NO2 |
|---|
| Mol. Mass. | 255.3117 |
|---|
| SMILES | Oc1cc2CCNCC(c3ccccc3)c2cc1O |
|---|
| Structure | 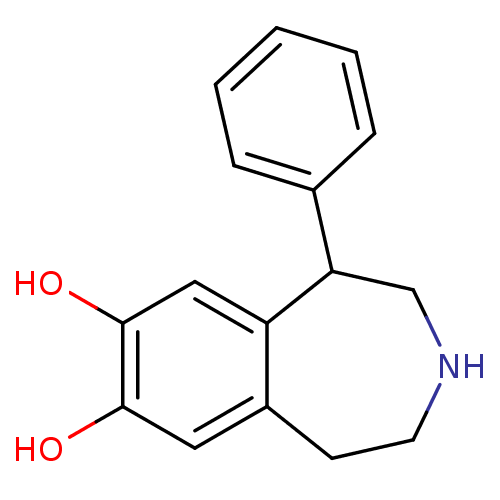
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Lebar, MD; Hahn, KN; Mutka, T; Maignan, P; McClintock, JB; Amsler, CD; van Olphen, A; Kyle, DE; Baker, BJ CNS and antimalarial activity of synthetic meridianin and psammopemmin analogs. Bioorg Med Chem19:5756-62 (2011) [PubMed] Article
Lebar, MD; Hahn, KN; Mutka, T; Maignan, P; McClintock, JB; Amsler, CD; van Olphen, A; Kyle, DE; Baker, BJ CNS and antimalarial activity of synthetic meridianin and psammopemmin analogs. Bioorg Med Chem19:5756-62 (2011) [PubMed] Article