| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Tyrosine-protein kinase SYK |
|---|
| Ligand | BDBM50355393 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_773606 (CHEMBL1839972) |
|---|
| IC50 | 5358±n/a nM |
|---|
| Citation |  Guagnano, V; Furet, P; Spanka, C; Bordas, V; Le Douget, M; Stamm, C; Brueggen, J; Jensen, MR; Schnell, C; Schmid, H; Wartmann, M; Berghausen, J; Drueckes, P; Zimmerlin, A; Bussiere, D; Murray, J; Graus Porta, D Discovery of 3-(2,6-dichloro-3,5-dimethoxy-phenyl)-1-{6-[4-(4-ethyl-piperazin-1-yl)-phenylamino]-pyrimidin-4-yl}-1-methyl-urea (NVP-BGJ398), a potent and selective inhibitor of the fibroblast growth factor receptor family of receptor tyrosine kinase. J Med Chem54:7066-83 (2011) [PubMed] Article Guagnano, V; Furet, P; Spanka, C; Bordas, V; Le Douget, M; Stamm, C; Brueggen, J; Jensen, MR; Schnell, C; Schmid, H; Wartmann, M; Berghausen, J; Drueckes, P; Zimmerlin, A; Bussiere, D; Murray, J; Graus Porta, D Discovery of 3-(2,6-dichloro-3,5-dimethoxy-phenyl)-1-{6-[4-(4-ethyl-piperazin-1-yl)-phenylamino]-pyrimidin-4-yl}-1-methyl-urea (NVP-BGJ398), a potent and selective inhibitor of the fibroblast growth factor receptor family of receptor tyrosine kinase. J Med Chem54:7066-83 (2011) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Tyrosine-protein kinase SYK |
|---|
| Name: | Tyrosine-protein kinase SYK |
|---|
| Synonyms: | KSYK_HUMAN | SYK | Spleen Tyrosine Kinase | Spleen tyrosine kinase (SYK) | Tyrosine-protein kinase SYK (Syk) |
|---|
| Type: | Enzyme |
|---|
| Mol. Mass.: | 72079.99 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | P43405 |
|---|
| Residue: | 635 |
|---|
| Sequence: | MASSGMADSANHLPFFFGNITREEAEDYLVQGGMSDGLYLLRQSRNYLGGFALSVAHGRK
AHHYTIERELNGTYAIAGGRTHASPADLCHYHSQESDGLVCLLKKPFNRPQGVQPKTGPF
EDLKENLIREYVKQTWNLQGQALEQAIISQKPQLEKLIATTAHEKMPWFHGKISREESEQ
IVLIGSKTNGKFLIRARDNNGSYALCLLHEGKVLHYRIDKDKTGKLSIPEGKKFDTLWQL
VEHYSYKADGLLRVLTVPCQKIGTQGNVNFGGRPQLPGSHPATWSAGGIISRIKSYSFPK
PGHRKSSPAQGNRQESTVSFNPYEPELAPWAADKGPQREALPMDTEVYESPYADPEEIRP
KEVYLDRKLLTLEDKELGSGNFGTVKKGYYQMKKVVKTVAVKILKNEANDPALKDELLAE
ANVMQQLDNPYIVRMIGICEAESWMLVMEMAELGPLNKYLQQNRHVKDKNIIELVHQVSM
GMKYLEESNFVHRDLAARNVLLVTQHYAKISDFGLSKALRADENYYKAQTHGKWPVKWYA
PECINYYKFSSKSDVWSFGVLMWEAFSYGQKPYRGMKGSEVTAMLEKGERMGCPAGCPRE
MYDLMNLCWTYDVENRPGFAAVELRLRNYYYDVVN
|
|
|
|---|
| BDBM50355393 |
|---|
| n/a |
|---|
| Name | BDBM50355393 |
|---|
| Synonyms: | BGJ398 | CHEMBL1834657 | US9434697, BGJ398 | US9730931, BGJ398 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C26H31Cl2N7O3 |
|---|
| Mol. Mass. | 560.475 |
|---|
| SMILES | CCN1CCN(CC1)c1ccc(Nc2cc(ncn2)N(C)C(=O)Nc2c(Cl)c(OC)cc(OC)c2Cl)cc1 |
|---|
| Structure | 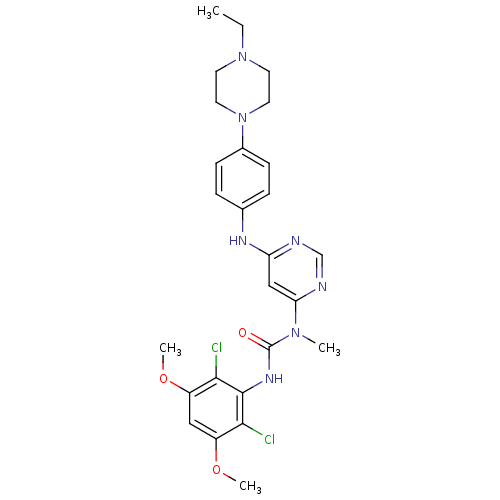
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Guagnano, V; Furet, P; Spanka, C; Bordas, V; Le Douget, M; Stamm, C; Brueggen, J; Jensen, MR; Schnell, C; Schmid, H; Wartmann, M; Berghausen, J; Drueckes, P; Zimmerlin, A; Bussiere, D; Murray, J; Graus Porta, D Discovery of 3-(2,6-dichloro-3,5-dimethoxy-phenyl)-1-{6-[4-(4-ethyl-piperazin-1-yl)-phenylamino]-pyrimidin-4-yl}-1-methyl-urea (NVP-BGJ398), a potent and selective inhibitor of the fibroblast growth factor receptor family of receptor tyrosine kinase. J Med Chem54:7066-83 (2011) [PubMed] Article
Guagnano, V; Furet, P; Spanka, C; Bordas, V; Le Douget, M; Stamm, C; Brueggen, J; Jensen, MR; Schnell, C; Schmid, H; Wartmann, M; Berghausen, J; Drueckes, P; Zimmerlin, A; Bussiere, D; Murray, J; Graus Porta, D Discovery of 3-(2,6-dichloro-3,5-dimethoxy-phenyl)-1-{6-[4-(4-ethyl-piperazin-1-yl)-phenylamino]-pyrimidin-4-yl}-1-methyl-urea (NVP-BGJ398), a potent and selective inhibitor of the fibroblast growth factor receptor family of receptor tyrosine kinase. J Med Chem54:7066-83 (2011) [PubMed] Article