| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Cytochrome P450 2D6 |
|---|
| Ligand | BDBM50355405 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_773773 (CHEMBL1840139) |
|---|
| IC50 | >75000±n/a nM |
|---|
| Citation |  Lim, J; Taoka, B; Otte, RD; Spencer, K; Dinsmore, CJ; Altman, MD; Chan, G; Rosenstein, C; Sharma, S; Su, HP; Szewczak, AA; Xu, L; Yin, H; Zugay-Murphy, J; Marshall, CG; Young, JR Discovery of 1-amino-5H-pyrido[4,3-b]indol-4-carboxamide inhibitors of Janus kinase 2 (JAK2) for the treatment of myeloproliferative disorders. J Med Chem54:7334-49 (2011) [PubMed] Article Lim, J; Taoka, B; Otte, RD; Spencer, K; Dinsmore, CJ; Altman, MD; Chan, G; Rosenstein, C; Sharma, S; Su, HP; Szewczak, AA; Xu, L; Yin, H; Zugay-Murphy, J; Marshall, CG; Young, JR Discovery of 1-amino-5H-pyrido[4,3-b]indol-4-carboxamide inhibitors of Janus kinase 2 (JAK2) for the treatment of myeloproliferative disorders. J Med Chem54:7334-49 (2011) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Cytochrome P450 2D6 |
|---|
| Name: | Cytochrome P450 2D6 |
|---|
| Synonyms: | CP2D6_HUMAN | CYP2D6 | CYP2DL1 | CYPIID6 | Cytochrome P450 2D6 (CYP2D6) | Debrisoquine 4-hydroxylase | P450-DB1 |
|---|
| Type: | Protein |
|---|
| Mol. Mass.: | 55774.82 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | P10635 |
|---|
| Residue: | 497 |
|---|
| Sequence: | MGLEALVPLAVIVAIFLLLVDLMHRRQRWAARYPPGPLPLPGLGNLLHVDFQNTPYCFDQ
LRRRFGDVFSLQLAWTPVVVLNGLAAVREALVTHGEDTADRPPVPITQILGFGPRSQGVF
LARYGPAWREQRRFSVSTLRNLGLGKKSLEQWVTEEAACLCAAFANHSGRPFRPNGLLDK
AVSNVIASLTCGRRFEYDDPRFLRLLDLAQEGLKEESGFLREVLNAVPVLLHIPALAGKV
LRFQKAFLTQLDELLTEHRMTWDPAQPPRDLTEAFLAEMEKAKGNPESSFNDENLRIVVA
DLFSAGMVTTSTTLAWGLLLMILHPDVQRRVQQEIDDVIGQVRRPEMGDQAHMPYTTAVI
HEVQRFGDIVPLGVTHMTSRDIEVQGFRIPKGTTLITNLSSVLKDEAVWEKPFRFHPEHF
LDAQGHFVKPEAFLPFSAGRRACLGEPLARMELFLFFTSLLQHFSFSVPTGQPRPSHHGV
FAFLVSPSPYELCAVPR
|
|
|
|---|
| BDBM50355405 |
|---|
| n/a |
|---|
| Name | BDBM50355405 |
|---|
| Synonyms: | CHEMBL1835153 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C21H18F3N7O |
|---|
| Mol. Mass. | 441.4091 |
|---|
| SMILES | NC(=O)c1cnc(N[C@H](C2CC2)C(F)(F)F)c2c3ccc(cc3[nH]c12)-c1cnc(N)nc1 |r| |
|---|
| Structure | 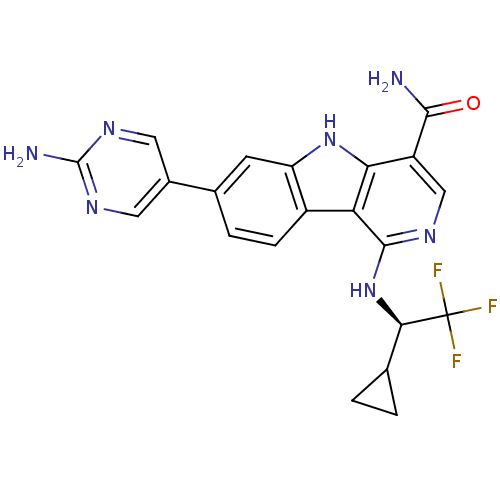
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Lim, J; Taoka, B; Otte, RD; Spencer, K; Dinsmore, CJ; Altman, MD; Chan, G; Rosenstein, C; Sharma, S; Su, HP; Szewczak, AA; Xu, L; Yin, H; Zugay-Murphy, J; Marshall, CG; Young, JR Discovery of 1-amino-5H-pyrido[4,3-b]indol-4-carboxamide inhibitors of Janus kinase 2 (JAK2) for the treatment of myeloproliferative disorders. J Med Chem54:7334-49 (2011) [PubMed] Article
Lim, J; Taoka, B; Otte, RD; Spencer, K; Dinsmore, CJ; Altman, MD; Chan, G; Rosenstein, C; Sharma, S; Su, HP; Szewczak, AA; Xu, L; Yin, H; Zugay-Murphy, J; Marshall, CG; Young, JR Discovery of 1-amino-5H-pyrido[4,3-b]indol-4-carboxamide inhibitors of Janus kinase 2 (JAK2) for the treatment of myeloproliferative disorders. J Med Chem54:7334-49 (2011) [PubMed] Article