| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Mitogen-activated protein kinase 10 |
|---|
| Ligand | BDBM50358497 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_789936 (CHEMBL1925289) |
|---|
| IC50 | 7±n/a nM |
|---|
| Citation |  Song, X; Chen, W; Lin, L; Ruiz, CH; Cameron, MD; Duckett, DR; Kamenecka, TM Synthesis and SAR of 2-phenoxypyridines as novel c-Jun N-terminal kinase inhibitors. Bioorg Med Chem Lett21:7072-5 (2011) [PubMed] Article Song, X; Chen, W; Lin, L; Ruiz, CH; Cameron, MD; Duckett, DR; Kamenecka, TM Synthesis and SAR of 2-phenoxypyridines as novel c-Jun N-terminal kinase inhibitors. Bioorg Med Chem Lett21:7072-5 (2011) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Mitogen-activated protein kinase 10 |
|---|
| Name: | Mitogen-activated protein kinase 10 |
|---|
| Synonyms: | JNK3 | JNK3A | MAP kinase p49 3F12 | MAPK10 | MK10_HUMAN | Mitogen-Activated Protein Kinase 10 (JNK3) | Mitogen-activated protein kinase 10 (Stress-activated protein kinase JNK3) (c-Jun N-terminal kinase 3) (MAP kinase p49 3F12) | Mitogen-activated protein kinase 10/Receptor-interacting serine/threonine-protein kinase 1 | PRKM10 | SAPK1B | Stress-activated protein kinase JNK3 | c-Jun N-terminal kinase 3 (JNK3) |
|---|
| Type: | Enzyme |
|---|
| Mol. Mass.: | 52586.89 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | n/a |
|---|
| Residue: | 464 |
|---|
| Sequence: | MSLHFLYYCSEPTLDVKIAFCQGFDKQVDVSYIAKHYNMSKSKVDNQFYSVEVGDSTFTV
LKRYQNLKPIGSGAQGIVCAAYDAVLDRNVAIKKLSRPFQNQTHAKRAYRELVLMKCVNH
KNIISLLNVFTPQKTLEEFQDVYLVMELMDANLCQVIQMELDHERMSYLLYQMLCGIKHL
HSAGIIHRDLKPSNIVVKSDCTLKILDFGLARTAGTSFMMTPYVVTRYYRAPEVILGMGY
KENVDIWSVGCIMGEMVRHKILFPGRDYIDQWNKVIEQLGTPCPEFMKKLQPTVRNYVEN
RPKYAGLTFPKLFPDSLFPADSEHNKLKASQARDLLSKMLVIDPAKRISVDDALQHPYIN
VWYDPAEVEAPPPQIYDKQLDEREHTIEEWKELIYKEVMNSEEKTKNGVVKGQPSPSGAA
VNSSESLPPSSSVNDISSMSTDQTLASDTDSSLEASAGPLGCCR
|
|
|
|---|
| BDBM50358497 |
|---|
| n/a |
|---|
| Name | BDBM50358497 |
|---|
| Synonyms: | CHEMBL1923217 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C18H15ClN4O3 |
|---|
| Mol. Mass. | 370.79 |
|---|
| SMILES | COc1ccc(Oc2ncc(Cl)c(Nc3ccccc3C(N)=O)n2)cc1 |
|---|
| Structure | 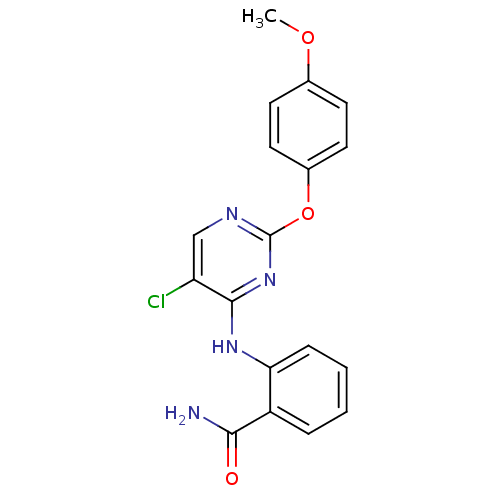
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Song, X; Chen, W; Lin, L; Ruiz, CH; Cameron, MD; Duckett, DR; Kamenecka, TM Synthesis and SAR of 2-phenoxypyridines as novel c-Jun N-terminal kinase inhibitors. Bioorg Med Chem Lett21:7072-5 (2011) [PubMed] Article
Song, X; Chen, W; Lin, L; Ruiz, CH; Cameron, MD; Duckett, DR; Kamenecka, TM Synthesis and SAR of 2-phenoxypyridines as novel c-Jun N-terminal kinase inhibitors. Bioorg Med Chem Lett21:7072-5 (2011) [PubMed] Article