| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Leukotriene B4 receptor 1 |
|---|
| Ligand | BDBM50280885 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEBML_98512 |
|---|
| Ki | 751±n/a nM |
|---|
| Citation |  Scott Sawyer, J; Baldwin, RF; Froelich, LL; Saussy, DL; Jackson, WT Synthesis and pharmacologic activity of hydroxyacetophenone-substituted benzophenone/xanthone leukotriene B4 receptor antagonists Bioorg Med Chem Lett3:1981-1984 (1993) Article Scott Sawyer, J; Baldwin, RF; Froelich, LL; Saussy, DL; Jackson, WT Synthesis and pharmacologic activity of hydroxyacetophenone-substituted benzophenone/xanthone leukotriene B4 receptor antagonists Bioorg Med Chem Lett3:1981-1984 (1993) Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Leukotriene B4 receptor 1 |
|---|
| Name: | Leukotriene B4 receptor 1 |
|---|
| Synonyms: | BLT | BLT1 | BLTR | CMKRL1 | Chemoattractant receptor-like 1 | G-protein coupled receptor 16 | GPR16 | LT4R1_HUMAN | LTB4-R 1 | LTB4R | Leukotriene B4 R1 | Leukotriene B4 receptor | Leukotriene B4 receptor 1 | P2RY7 | P2Y purinoceptor 7 | P2Y7 |
|---|
| Type: | Enzyme Catalytic Domain |
|---|
| Mol. Mass.: | 37582.68 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | Q15722 |
|---|
| Residue: | 352 |
|---|
| Sequence: | MNTTSSAAPPSLGVEFISLLAIILLSVALAVGLPGNSFVVWSILKRMQKRSVTALMVLNL
ALADLAVLLTAPFFLHFLAQGTWSFGLAGCRLCHYVCGVSMYASVLLITAMSLDRSLAVA
RPFVSQKLRTKAMARRVLAGIWVLSFLLATPVLAYRTVVPWKTNMSLCFPRYPSEGHRAF
HLIFEAVTGFLLPFLAVVASYSDIGRRLQARRFRRSRRTGRLVVLIILTFAAFWLPYHVV
NLAEAGRALAGQAAGLGLVGKRLSLARNVLIALAFLSSSVNPVLYACAGGGLLRSAGVGF
VAKLLEGTGSEASSTRRGGSLGQTARSGPAALEPGPSESLTASSPLKLNELN
|
|
|
|---|
| BDBM50280885 |
|---|
| n/a |
|---|
| Name | BDBM50280885 |
|---|
| Synonyms: | CHEMBL58534 | disodium 6-[3-(4-acetyl-2-ethyl-5-olatophenoxy)propoxy]-9-oxo-9H-2-xanthenecarboxylate |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C27H22O8 |
|---|
| Mol. Mass. | 474.4599 |
|---|
| SMILES | CCc1cc(C(C)=O)c([O-])cc1OCCCOc1ccc2c(c1)oc1ccc(cc1c2=O)C([O-])=O |
|---|
| Structure | 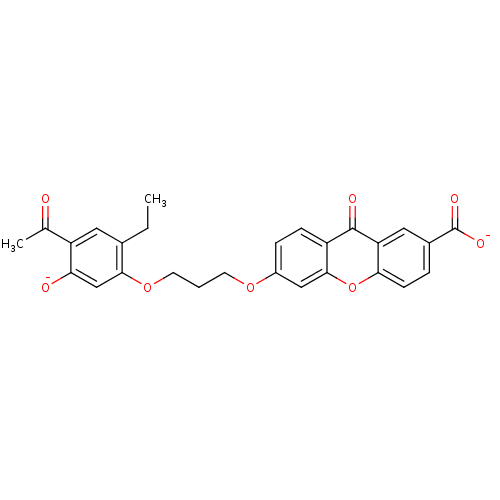
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Scott Sawyer, J; Baldwin, RF; Froelich, LL; Saussy, DL; Jackson, WT Synthesis and pharmacologic activity of hydroxyacetophenone-substituted benzophenone/xanthone leukotriene B4 receptor antagonists Bioorg Med Chem Lett3:1981-1984 (1993) Article
Scott Sawyer, J; Baldwin, RF; Froelich, LL; Saussy, DL; Jackson, WT Synthesis and pharmacologic activity of hydroxyacetophenone-substituted benzophenone/xanthone leukotriene B4 receptor antagonists Bioorg Med Chem Lett3:1981-1984 (1993) Article