| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Angiotensin-converting enzyme 2 |
|---|
| Ligand | BDBM50283658 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_35588 (CHEMBL876577) |
|---|
| IC50 | 22±n/a nM |
|---|
| Citation |  De Lombaert, S; Tan, J; Stamford, LJ; Sakane, Y; Berry, C; Ghai, RD Dual inhibition of neutral endopeptidase and angiotensin-converting enzyme by N-phosphonomethyl and N-carboxyalkyl dipeptides Bioorg Med Chem Lett4:2715-2720 (1994) Article De Lombaert, S; Tan, J; Stamford, LJ; Sakane, Y; Berry, C; Ghai, RD Dual inhibition of neutral endopeptidase and angiotensin-converting enzyme by N-phosphonomethyl and N-carboxyalkyl dipeptides Bioorg Med Chem Lett4:2715-2720 (1994) Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Angiotensin-converting enzyme 2 |
|---|
| Name: | Angiotensin-converting enzyme 2 |
|---|
| Synonyms: | ACEI-ACE2 | Angiotensin-converting enzyme |
|---|
| Type: | n/a |
|---|
| Mol. Mass.: | n/a |
|---|
| Description: | ASSAY_ID of EBI is 35589 |
|---|
| Components: | This complex has 2 components. |
|---|
| Component 1 |
| Name: | Angiotensin-converting enzyme |
|---|
| Synonyms: | ACE | ACE_HUMAN | Angiotensin converting enzyme (ACE) | Angiotensin-converting enzyme, ACE | Angiotensin-converting enzyme, soluble form | Angiotensin-converting enzyme, somatic isoform | CD_antigen=CD143 | DCP | DCP1 | Dipeptidyl carboxypeptidase I | Kininase II |
|---|
| Type: | Enzyme |
|---|
| Mol. Mass.: | 149709.01 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | n/a |
|---|
| Residue: | 1306 |
|---|
| Sequence: | MGAASGRRGPGLLLPLPLLLLLPPQPALALDPGLQPGNFSADEAGAQLFAQSYNSSAEQV
LFQSVAASWAHDTNITAENARRQEEAALLSQEFAEAWGQKAKELYEPIWQNFTDPQLRRI
IGAVRTLGSANLPLAKRQQYNALLSNMSRIYSTAKVCLPNKTATCWSLDPDLTNILASSR
SYAMLLFAWEGWHNAAGIPLKPLYEDFTALSNEAYKQDGFTDTGAYWRSWYNSPTFEDDL
EHLYQQLEPLYLNLHAFVRRALHRRYGDRYINLRGPIPAHLLGDMWAQSWENIYDMVVPF
PDKPNLDVTSTMLQQGWNATHMFRVAEEFFTSLELSPMPPEFWEGSMLEKPADGREVVCH
ASAWDFYNRKDFRIKQCTRVTMDQLSTVHHEMGHIQYYLQYKDLPVSLRRGANPGFHEAI
GDVLALSVSTPEHLHKIGLLDRVTNDTESDINYLLKMALEKIAFLPFGYLVDQWRWGVFS
GRTPPSRYNFDWWYLRTKYQGICPPVTRNETHFDAGAKFHVPNVTPYIRYFVSFVLQFQF
HEALCKEAGYEGPLHQCDIYRSTKAGAKLRKVLQAGSSRPWQEVLKDMVGLDALDAQPLL
KYFQPVTQWLQEQNQQNGEVLGWPEYQWHPPLPDNYPEGIDLVTDEAEASKFVEEYDRTS
QVVWNEYAEANWNYNTNITTETSKILLQKNMQIANHTLKYGTQARKFDVNQLQNTTIKRI
IKKVQDLERAALPAQELEEYNKILLDMETTYSVATVCHPNGSCLQLEPDLTNVMATSRKY
EDLLWAWEGWRDKAGRAILQFYPKYVELINQAARLNGYVDAGDSWRSMYETPSLEQDLER
LFQELQPLYLNLHAYVRRALHRHYGAQHINLEGPIPAHLLGNMWAQTWSNIYDLVVPFPS
APSMDTTEAMLKQGWTPRRMFKEADDFFTSLGLLPVPPEFWNKSMLEKPTDGREVVCHAS
AWDFYNGKDFRIKQCTTVNLEDLVVAHHEMGHIQYFMQYKDLPVALREGANPGFHEAIGD
VLALSVSTPKHLHSLNLLSSEGGSDEHDINFLMKMALDKIAFIPFSYLVDQWRWRVFDGS
ITKENYNQEWWSLRLKYQGLCPPVPRTQGDFDPGAKFHIPSSVPYIRYFVSFIIQFQFHE
ALCQAAGHTGPLHKCDIYQSKEAGQRLATAMKLGFSRPWPEAMQLITGQPNMSASAMLSY
FKPLLDWLRTENELHGEKLGWPQYNWTPNSARSEGPLPDSGRVSFLGLDLDAQQARVGQW
LLLFLGIALLVATLGLSQRLFSIRHRSLHRHSHGPQFGSEVELRHS
|
|
|
|---|
| Component 2 |
| Name: | Angiotensin-converting enzyme 2 |
|---|
| Synonyms: | ACE-related carboxypeptidase | ACE2 | ACE2_HUMAN | ACEH | Angiotensin-converting enzyme homolog | Angiotensin-converting enzyme-related carboxypeptidase | Metalloprotease MPROT15 |
|---|
| Type: | Enzyme |
|---|
| Mol. Mass.: | 92448.86 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | n/a |
|---|
| Residue: | 805 |
|---|
| Sequence: | MSSSSWLLLSLVAVTAAQSTIEEQAKTFLDKFNHEAEDLFYQSSLASWNYNTNITEENVQ
NMNNAGDKWSAFLKEQSTLAQMYPLQEIQNLTVKLQLQALQQNGSSVLSEDKSKRLNTIL
NTMSTIYSTGKVCNPDNPQECLLLEPGLNEIMANSLDYNERLWAWESWRSEVGKQLRPLY
EEYVVLKNEMARANHYEDYGDYWRGDYEVNGVDGYDYSRGQLIEDVEHTFEEIKPLYEHL
HAYVRAKLMNAYPSYISPIGCLPAHLLGDMWGRFWTNLYSLTVPFGQKPNIDVTDAMVDQ
AWDAQRIFKEAEKFFVSVGLPNMTQGFWENSMLTDPGNVQKAVCHPTAWDLGKGDFRILM
CTKVTMDDFLTAHHEMGHIQYDMAYAAQPFLLRNGANEGFHEAVGEIMSLSAATPKHLKS
IGLLSPDFQEDNETEINFLLKQALTIVGTLPFTYMLEKWRWMVFKGEIPKDQWMKKWWEM
KREIVGVVEPVPHDETYCDPASLFHVSNDYSFIRYYTRTLYQFQFQEALCQAAKHEGPLH
KCDISNSTEAGQKLFNMLRLGKSEPWTLALENVVGAKNMNVRPLLNYFEPLFTWLKDQNK
NSFVGWSTDWSPYADQSIKVRISLKSALGDKAYEWNDNEMYLFRSSVAYAMRQYFLKVKN
QMILFGEEDVRVANLKPRISFNFFVTAPKNVSDIIPRTEVEKAIRMSRSRINDAFRLNDN
SLEFLGIQPTLGPPNQPPVSIWLIVFGVVMGVIVVGIVILIFTGIRDRKKKNKARSGENP
YASIDISKGENNPGFQNTDDVQTSF
|
|
|
|---|
| BDBM50283658 |
|---|
| n/a |
|---|
| Name | BDBM50283658 |
|---|
| Synonyms: | (S)-3-Biphenyl-4-yl-2-[(S)-2-((S)-1-carboxy-ethylamino)-3-methyl-pentanoylamino]-propionic acid | CHEMBL94173 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C24H30N2O5 |
|---|
| Mol. Mass. | 426.5054 |
|---|
| SMILES | CC[C@H](C)C(N[C@@H](C)C(O)=O)C(=O)N[C@@H](Cc1ccc(cc1)-c1ccccc1)C(O)=O |
|---|
| Structure | 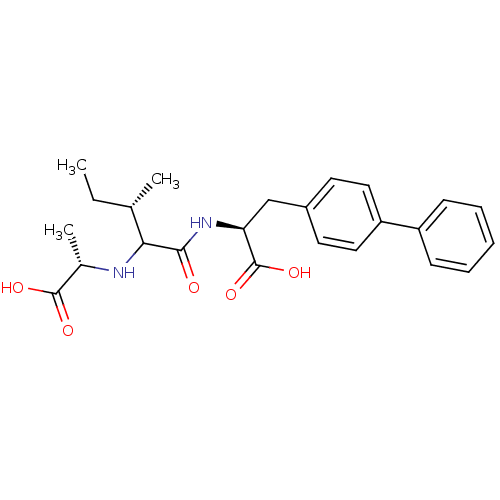
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 De Lombaert, S; Tan, J; Stamford, LJ; Sakane, Y; Berry, C; Ghai, RD Dual inhibition of neutral endopeptidase and angiotensin-converting enzyme by N-phosphonomethyl and N-carboxyalkyl dipeptides Bioorg Med Chem Lett4:2715-2720 (1994) Article
De Lombaert, S; Tan, J; Stamford, LJ; Sakane, Y; Berry, C; Ghai, RD Dual inhibition of neutral endopeptidase and angiotensin-converting enzyme by N-phosphonomethyl and N-carboxyalkyl dipeptides Bioorg Med Chem Lett4:2715-2720 (1994) Article