| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Neuromedin-B receptor |
|---|
| Ligand | BDBM50071733 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEBML_39622 |
|---|
| Ki | 9.1±n/a nM |
|---|
| Citation |  Ashwood, V; Brownhill, V; Higginbottom, M; Horwell, DC; Hughes, J; Lewthwaite, RA; McKnight, AT; Pinnock, RD; Pritchard, MC; Suman-Chauhan, N; Webb, C; Williams, SC PD 176252--the first high affinity non-peptide gastrin-releasing peptide (BB2) receptor antagonist. Bioorg Med Chem Lett8:2589-94 (1999) [PubMed] Ashwood, V; Brownhill, V; Higginbottom, M; Horwell, DC; Hughes, J; Lewthwaite, RA; McKnight, AT; Pinnock, RD; Pritchard, MC; Suman-Chauhan, N; Webb, C; Williams, SC PD 176252--the first high affinity non-peptide gastrin-releasing peptide (BB2) receptor antagonist. Bioorg Med Chem Lett8:2589-94 (1999) [PubMed] |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Neuromedin-B receptor |
|---|
| Name: | Neuromedin-B receptor |
|---|
| Synonyms: | Bombesin 1 | NMB-R | NMBR | NMBR_HUMAN | Neuromedin B | Neuromedin B receptor | Neuromedin-B receptor | Neuromedin-B-preferring bombesin receptor |
|---|
| Type: | Enzyme Catalytic Domain |
|---|
| Mol. Mass.: | 43447.11 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | Bombesin 1 NMBR 0::P28336 |
|---|
| Residue: | 390 |
|---|
| Sequence: | MPSKSLSNLSVTTGANESGSVPEGWERDFLPASDGTTTELVIRCVIPSLYLLIITVGLLG
NIMLVKIFITNSAMRSVPNIFISNLAAGDLLLLLTCVPVDASRYFFDEWMFGKVGCKLIP
VIQLTSVGVSVFTLTALSADRYRAIVNPMDMQTSGALLRTCVKAMGIWVVSVLLAVPEAV
FSEVARISSLDNSSFTACIPYPQTDELHPKIHSVLIFLVYFLIPLAIISIYYYHIAKTLI
KSAHNLPGEYNEHTKKQMETRKRLAKIVLVFVGCFIFCWFPNHILYMYRSFNYNEIDPSL
GHMIVTLVARVLSFGNSCVNPFALYLLSESFRRHFNSQLCCGRKSYQERGTSYLLSSSAV
RMTSLKSNAKNMVTNSVLLNGHSMKQEMAL
|
|
|
|---|
| BDBM50071733 |
|---|
| n/a |
|---|
| Name | BDBM50071733 |
|---|
| Synonyms: | CHEMBL413196 | Compound GRP |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C130H203N37O32S2 |
|---|
| Mol. Mass. | 2860.362 |
|---|
| SMILES | CSCC[C@H](NC(=O)[C@H](CC(C)C)NC(=O)[C@H](Cc1cnc[nH]1)NC(=O)CNC(=O)[C@@H](NC(=O)[C@H](C)NC(=O)[C@H](Cc1c[nH]c2ccccc12)NC(=O)[C@H](Cc1cnc[nH]1)NC(=O)[C@H](CC(N)=O)NC(=O)CNC(=O)[C@H](CCCNC(N)=N)NC(=O)[C@@H]1CCCN1C(=O)[C@H](Cc1ccc(O)cc1)NC(=O)[C@H](CCSC)NC(=O)[C@H](CCCCN)NC(=O)[C@@H](NC(=O)[C@H](CC(C)C)NC(=O)[C@@H](NC(=O)[C@@H](NC(=O)CNC(=O)CNC(=O)CNC(=O)[C@H](C)NC(=O)[C@@H]1CCCN1C(=O)[C@H](CC(C)C)NC(=O)[C@@H]1CCCN1C(=O)[C@@H](N)C(C)C)[C@@H](C)O)C(C)C)[C@@H](C)O)C(C)C)C(O)=O |
|---|
| Structure | 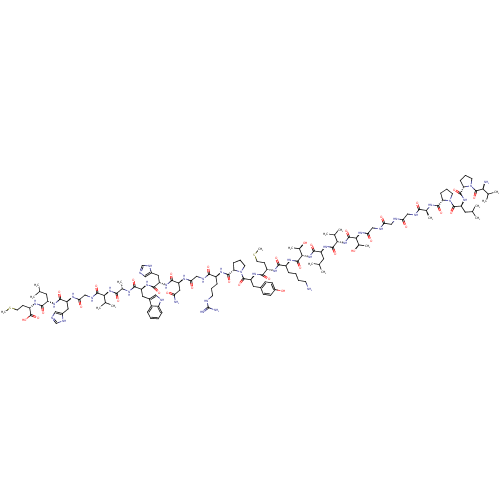
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Ashwood, V; Brownhill, V; Higginbottom, M; Horwell, DC; Hughes, J; Lewthwaite, RA; McKnight, AT; Pinnock, RD; Pritchard, MC; Suman-Chauhan, N; Webb, C; Williams, SC PD 176252--the first high affinity non-peptide gastrin-releasing peptide (BB2) receptor antagonist. Bioorg Med Chem Lett8:2589-94 (1999) [PubMed]
Ashwood, V; Brownhill, V; Higginbottom, M; Horwell, DC; Hughes, J; Lewthwaite, RA; McKnight, AT; Pinnock, RD; Pritchard, MC; Suman-Chauhan, N; Webb, C; Williams, SC PD 176252--the first high affinity non-peptide gastrin-releasing peptide (BB2) receptor antagonist. Bioorg Med Chem Lett8:2589-94 (1999) [PubMed]