| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Estrogen receptor |
|---|
| Ligand | BDBM19441 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_67488 (CHEMBL679325) |
|---|
| IC50 | 2±n/a nM |
|---|
| Citation |  Miller, CP; Jirkovsky, I; Tran, BD; Harris, HA; Moran, RA; Komm, BS Synthesis and estrogenic activities of novel 7-thiosubstituted estratriene derivatives. Bioorg Med Chem Lett10:147-51 (2000) [PubMed] Miller, CP; Jirkovsky, I; Tran, BD; Harris, HA; Moran, RA; Komm, BS Synthesis and estrogenic activities of novel 7-thiosubstituted estratriene derivatives. Bioorg Med Chem Lett10:147-51 (2000) [PubMed] |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Estrogen receptor |
|---|
| Name: | Estrogen receptor |
|---|
| Synonyms: | ER | ER-alpha | ESR | ESR1 | ESR1_HUMAN | Estradiol receptor | Estrogen receptor | Estrogen receptor (ER alpha) | Estrogen receptor (ER-alpha) | Estrogen receptor alpha (ER alpha) | Estrogen receptor alpha (ER) | NR3A1 | Nuclear receptor subfamily 3 group A member 1 |
|---|
| Type: | Protein |
|---|
| Mol. Mass.: | 66230.44 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | P03372 |
|---|
| Residue: | 595 |
|---|
| Sequence: | MTMTLHTKASGMALLHQIQGNELEPLNRPQLKIPLERPLGEVYLDSSKPAVYNYPEGAAY
EFNAAAAANAQVYGQTGLPYGPGSEAAAFGSNGLGGFPPLNSVSPSPLMLLHPPPQLSPF
LQPHGQQVPYYLENEPSGYTVREAGPPAFYRPNSDNRRQGGRERLASTNDKGSMAMESAK
ETRYCAVCNDYASGYHYGVWSCEGCKAFFKRSIQGHNDYMCPATNQCTIDKNRRKSCQAC
RLRKCYEVGMMKGGIRKDRRGGRMLKHKRQRDDGEGRGEVGSAGDMRAANLWPSPLMIKR
SKKNSLALSLTADQMVSALLDAEPPILYSEYDPTRPFSEASMMGLLTNLADRELVHMINW
AKRVPGFVDLTLHDQVHLLECAWLEILMIGLVWRSMEHPGKLLFAPNLLLDRNQGKCVEG
MVEIFDMLLATSSRFRMMNLQGEEFVCLKSIILLNSGVYTFLSSTLKSLEEKDHIHRVLD
KITDTLIHLMAKAGLTLQQQHQRLAQLLLILSHIRHMSNKGMEHLYSMKCKNVVPLYDLL
LEMLDAHRLHAPTSRGGASVEETDQSHLATAGSTSSHSLQKYYITGEAEGFPATV
|
|
|
|---|
| BDBM19441 |
|---|
| n/a |
|---|
| Name | BDBM19441 |
|---|
| Synonyms: | 2-(4-hydroxyphenyl)-3-({4-[2-(piperidin-1-yl)ethoxy]phenyl}carbonyl)-1-benzothiophen-6-ol | CHEMBL81 | Evista | Keoxifene | RALOXIFENE HYDROCHLORIDE | Raloxifene | Raloxifene (7) | Raloxifene, 6 | [6-hydroxy-2-(4-hydroxyphenyl)-1-benzothiophen-3-yl]-[4-(2-piperidin-1-ylethoxy)phenyl]methanone | cid_11071264 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C28H27NO4S |
|---|
| Mol. Mass. | 473.583 |
|---|
| SMILES | Oc1ccc(cc1)-c1sc2cc(O)ccc2c1C(=O)c1ccc(OCCN2CCCCC2)cc1 |
|---|
| Structure | 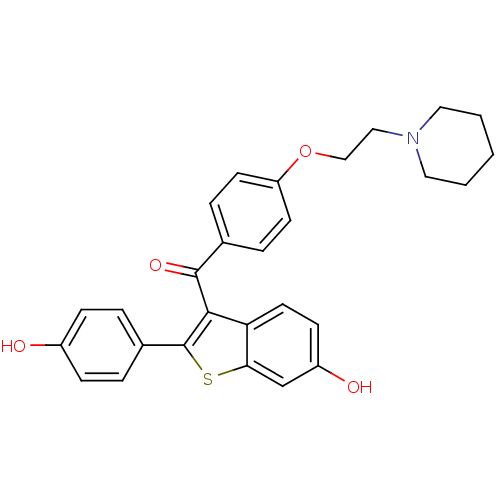
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Miller, CP; Jirkovsky, I; Tran, BD; Harris, HA; Moran, RA; Komm, BS Synthesis and estrogenic activities of novel 7-thiosubstituted estratriene derivatives. Bioorg Med Chem Lett10:147-51 (2000) [PubMed]
Miller, CP; Jirkovsky, I; Tran, BD; Harris, HA; Moran, RA; Komm, BS Synthesis and estrogenic activities of novel 7-thiosubstituted estratriene derivatives. Bioorg Med Chem Lett10:147-51 (2000) [PubMed]