| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Mu-type opioid receptor |
|---|
| Ligand | BDBM50009142 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEBML_146238 |
|---|
| IC50 | 420±n/a nM |
|---|
| Citation |  Salvadori, S; Marastoni, M; Balboni, G; Borea, PA; Morari, M; Tomatis, R Synthesis and structure-activity relationships of deltorphin analogues. J Med Chem34:1656-61 (1991) [PubMed] Salvadori, S; Marastoni, M; Balboni, G; Borea, PA; Morari, M; Tomatis, R Synthesis and structure-activity relationships of deltorphin analogues. J Med Chem34:1656-61 (1991) [PubMed] |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Mu-type opioid receptor |
|---|
| Name: | Mu-type opioid receptor |
|---|
| Synonyms: | M-OR-1 | MOR-1 | Mu opioid receptor | Mu-type opioid receptor (Mu) | OPIATE Mu | OPRM1 | OPRM_CAVPO |
|---|
| Type: | Enzyme Catalytic Domain |
|---|
| Mol. Mass.: | 11165.58 |
|---|
| Organism: | GUINEA PIG |
|---|
| Description: | P97266 |
|---|
| Residue: | 98 |
|---|
| Sequence: | YTKMKTATNIYIFNLALADALATSTLPFQSVNYLMGTWPFGTILCKIVISIDYYNMFTSI
FTLCTMSVDRYIAVCHPVKALDFRTPRNAKTVNVCNWI
|
|
|
|---|
| BDBM50009142 |
|---|
| n/a |
|---|
| Name | BDBM50009142 |
|---|
| Synonyms: | (S)-3-((S)-2-{(S)-2-[(S)-2-Amino-3-(4-hydroxy-phenyl)-propionylamino]-propionylamino}-3-phenyl-propionylamino)-N-{(S)-1-[(S)-1-(carbamoylmethyl-carbamoyl)-2-methyl-propylcarbamoyl]-2-methyl-propyl}-succinamic acid | 3-(2-{2-[2-Amino-3-(4-hydroxy-phenyl)-propionylamino]-propionylamino}-3-phenyl-propionylamino)-N-{1-[1-(carbamoylmethyl-carbamoyl)-2-methyl-propylcarbamoyl]-2-methyl-propyl}-succinamic acid | 3-(2-{2-[2-Amino-3-(4-hydroxy-phenyl)-propionylamino]-propionylamino}-3-phenyl-propionylamino)-N-{1-[1-(carbamoylmethyl-carbamoyl)-2-methyl-propylcarbamoyl]-2-methyl-propyl}-succinamic acid (deltorphin C) | 3-[1-[1-[1-amino-2-(4-hydroxyphenyl)-(1S)-ethylcarboxamido]-(1S)-ethylcarboxamido]-2-phenyl-(1S)-ethylcarboxamido]-3-[1-[1-carbamoylmethylcarbamoyl-2-methyl-(1S)-propylcarbamoyl]-2-methyl-(1S)-propylcarbamoyl]propanoic acid | CHEMBL281140 | Tyr-D-Ala-Phe-Asp-Val-Val-Gly-NH2 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C37H52N8O10 |
|---|
| Mol. Mass. | 768.8564 |
|---|
| SMILES | CC(C)[C@H](NC(=O)[C@@H](NC(=O)[C@H](CC(O)=O)NC(=O)[C@H](Cc1ccccc1)NC(=O)[C@H](C)NC(=O)[C@@H](N)Cc1ccc(O)cc1)C(C)C)C(=O)NCC(N)=O |
|---|
| Structure | 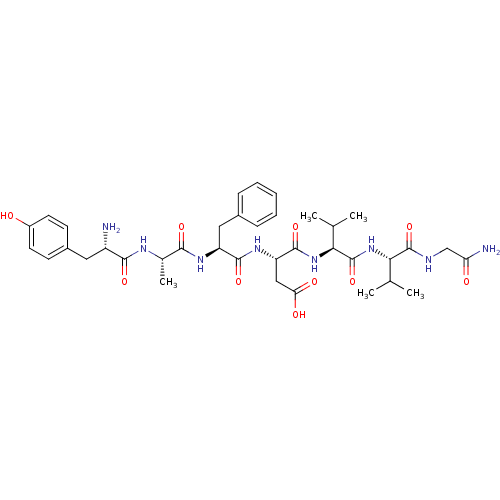
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Salvadori, S; Marastoni, M; Balboni, G; Borea, PA; Morari, M; Tomatis, R Synthesis and structure-activity relationships of deltorphin analogues. J Med Chem34:1656-61 (1991) [PubMed]
Salvadori, S; Marastoni, M; Balboni, G; Borea, PA; Morari, M; Tomatis, R Synthesis and structure-activity relationships of deltorphin analogues. J Med Chem34:1656-61 (1991) [PubMed]