| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Sodium-dependent dopamine transporter |
|---|
| Ligand | BDBM22165 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_62641 (CHEMBL676686) |
|---|
| Ki | 0.880000±n/a nM |
|---|
| Citation |  Goodman, MM; Kung, MP; Kabalka, GW; Kung, HF; Switzer, R Synthesis and characterization of radioiodinated N-(3-iodopropen-1-yl)-2 beta-carbomethoxy-3 beta-(4-chlorophenyl)tropanes: potential dopamine reuptake site imaging agents. J Med Chem37:1535-42 (1994) [PubMed] Goodman, MM; Kung, MP; Kabalka, GW; Kung, HF; Switzer, R Synthesis and characterization of radioiodinated N-(3-iodopropen-1-yl)-2 beta-carbomethoxy-3 beta-(4-chlorophenyl)tropanes: potential dopamine reuptake site imaging agents. J Med Chem37:1535-42 (1994) [PubMed] |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Sodium-dependent dopamine transporter |
|---|
| Name: | Sodium-dependent dopamine transporter |
|---|
| Synonyms: | DA transporter | Monoamine transporters; Norepininephrine & dopamine | SC6A3_RAT | Slc6a3 | Sodium-dependent dopamine transporter | Sodium-dependent dopamine transporter (DAT) |
|---|
| Type: | Multi-pass membrane protein |
|---|
| Mol. Mass.: | 68749.45 |
|---|
| Organism: | Rattus norvegicus (rat) |
|---|
| Description: | P23977 |
|---|
| Residue: | 619 |
|---|
| Sequence: | MSKSKCSVGPMSSVVAPAKESNAVGPREVELILVKEQNGVQLTNSTLINPPQTPVEAQER
ETWSKKIDFLLSVIGFAVDLANVWRFPYLCYKNGGGAFLVPYLLFMVIAGMPLFYMELAL
GQFNREGAAGVWKICPVLKGVGFTVILISFYVGFFYNVIIAWALHYFFSSFTMDLPWIHC
NNTWNSPNCSDAHASNSSDGLGLNDTFGTTPAAEYFERGVLHLHQSRGIDDLGPPRWQLT
ACLVLVIVLLYFSLWKGVKTSGKVVWITATMPYVVLTALLLRGVTLPGAMDGIRAYLSVD
FYRLCEASVWIDAATQVCFSLGVGFGVLIAFSSYNKFTNNCYRDAIITTSINSLTSFSSG
FVVFSFLGYMAQKHNVPIRDVATDGPGLIFIIYPEAIATLPLSSAWAAVFFLMLLTLGID
SAMGGMESVITGLVDEFQLLHRHRELFTLGIVLATFLLSLFCVTNGGIYVFTLLDHFAAG
TSILFGVLIEAIGVAWFYGVQQFSDDIKQMTGQRPNLYWRLCWKLVSPCFLLYVVVVSIV
TFRPPHYGAYIFPDWANALGWIIATSSMAMVPIYATYKFCSLPGSFREKLAYAITPEKDH
QLVDRGEVRQFTLRHWLLL
|
|
|
|---|
| BDBM22165 |
|---|
| n/a |
|---|
| Name | BDBM22165 |
|---|
| Synonyms: | 1-{2-[bis(4-fluorophenyl)methoxy]ethyl}-4-(3-phenylpropyl)piperazine | CHEMBL153260 | CHEMBL281594 | CHEMBL542933 | CHEMBL543876 | GBR 12909 | GBR-12909 | NCGC00015300 | US9944618, Compound ID No. 175 | Vanoxerine |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C28H32F2N2O |
|---|
| Mol. Mass. | 450.5633 |
|---|
| SMILES | Fc1ccc(cc1)C(OCCN1CCN(CCCc2ccccc2)CC1)c1ccc(F)cc1 |
|---|
| Structure | 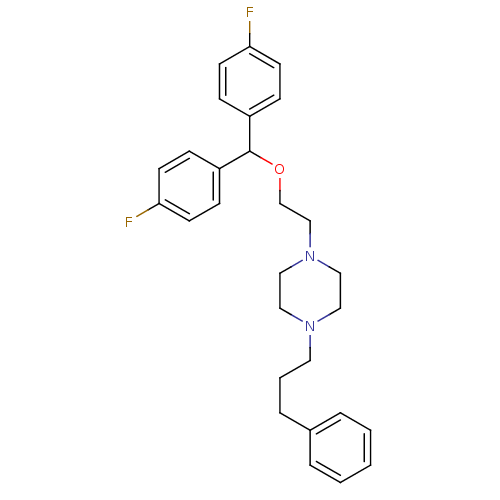
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Goodman, MM; Kung, MP; Kabalka, GW; Kung, HF; Switzer, R Synthesis and characterization of radioiodinated N-(3-iodopropen-1-yl)-2 beta-carbomethoxy-3 beta-(4-chlorophenyl)tropanes: potential dopamine reuptake site imaging agents. J Med Chem37:1535-42 (1994) [PubMed]
Goodman, MM; Kung, MP; Kabalka, GW; Kung, HF; Switzer, R Synthesis and characterization of radioiodinated N-(3-iodopropen-1-yl)-2 beta-carbomethoxy-3 beta-(4-chlorophenyl)tropanes: potential dopamine reuptake site imaging agents. J Med Chem37:1535-42 (1994) [PubMed]