| Reaction Details |
|---|
 | Report a problem with these data |
| Target | P-selectin |
|---|
| Ligand | BDBM50450369 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_200030 (CHEMBL810719) |
|---|
| IC50 | >1000000±n/a nM |
|---|
| Citation |  Wada, Y; Saito, T; Matsuda, N; Ohmoto, H; Yoshino, K; Ohashi, M; Kondo, H; Ishida, H; Kiso, M; Hasegawa, A Studies on selectin blockers. 2. Novel selectin blocker as potential therapeutics for inflammatory disorders. J Med Chem39:2055-9 (1996) [PubMed] Article Wada, Y; Saito, T; Matsuda, N; Ohmoto, H; Yoshino, K; Ohashi, M; Kondo, H; Ishida, H; Kiso, M; Hasegawa, A Studies on selectin blockers. 2. Novel selectin blocker as potential therapeutics for inflammatory disorders. J Med Chem39:2055-9 (1996) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| P-selectin |
|---|
| Name: | P-selectin |
|---|
| Synonyms: | GMRP | GRMP | LYAM3_HUMAN | P-selectin | P-selectin/P-selectin glycoprotein ligand 1 | SELP |
|---|
| Type: | PROTEIN |
|---|
| Mol. Mass.: | 90834.61 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | ChEMBL_1438999 |
|---|
| Residue: | 830 |
|---|
| Sequence: | MANCQIAILYQRFQRVVFGISQLLCFSALISELTNQKEVAAWTYHYSTKAYSWNISRKYC
QNRYTDLVAIQNKNEIDYLNKVLPYYSSYYWIGIRKNNKTWTWVGTKKALTNEAENWADN
EPNNKRNNEDCVEIYIKSPSAPGKWNDEHCLKKKHALCYTASCQDMSCSKQGECLETIGN
YTCSCYPGFYGPECEYVRECGELELPQHVLMNCSHPLGNFSFNSQCSFHCTDGYQVNGPS
KLECLASGIWTNKPPQCLAAQCPPLKIPERGNMTCLHSAKAFQHQSSCSFSCEEGFALVG
PEVVQCTASGVWTAPAPVCKAVQCQHLEAPSEGTMDCVHPLTAFAYGSSCKFECQPGYRV
RGLDMLRCIDSGHWSAPLPTCEAISCEPLESPVHGSMDCSPSLRAFQYDTNCSFRCAEGF
MLRGADIVRCDNLGQWTAPAPVCQALQCQDLPVPNEARVNCSHPFGAFRYQSVCSFTCNE
GLLLVGASVLQCLATGNWNSVPPECQAIPCTPLLSPQNGTMTCVQPLGSSSYKSTCQFIC
DEGYSLSGPERLDCTRSGRWTDSPPMCEAIKCPELFAPEQGSLDCSDTRGEFNVGSTCHF
SCDNGFKLEGPNNVECTTSGRWSATPPTCKGIASLPTPGLQCPALTTPGQGTMYCRHHPG
TFGFNTTCYFGCNAGFTLIGDSTLSCRPSGQWTAVTPACRAVKCSELHVNKPIAMNCSNL
WGNFSYGSICSFHCLEGQLLNGSAQTACQENGHWSTTVPTCQAGPLTIQEALTYFGGAVA
STIGLIMGGTLLALLRKRFRQKDDGKCPLNPHSHLGTYGVFTNAAFDPSP
|
|
|
|---|
| BDBM50450369 |
|---|
| n/a |
|---|
| Name | BDBM50450369 |
|---|
| Synonyms: | SIALYL LEWIS X | Sialyl LeX | Sialyl lewis-x | sLex | sialyl LewisX |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C31H52N2O23 |
|---|
| Mol. Mass. | 820.7442 |
|---|
| SMILES | [H][C@]1(O[C@@](C[C@H](O)[C@H]1NC(C)=O)(O[C@H]1[C@@H](O)[C@@H](CO)O[C@@H](O[C@H]([C@H](O)CO)[C@H](O[C@@H]2O[C@@H](C)[C@@H](O)[C@@H](O)[C@@H]2O)[C@@H](NC(C)=O)C=O)[C@@H]1O)C(O)=O)[C@H](O)[C@H](O)CO |
|---|
| Structure | 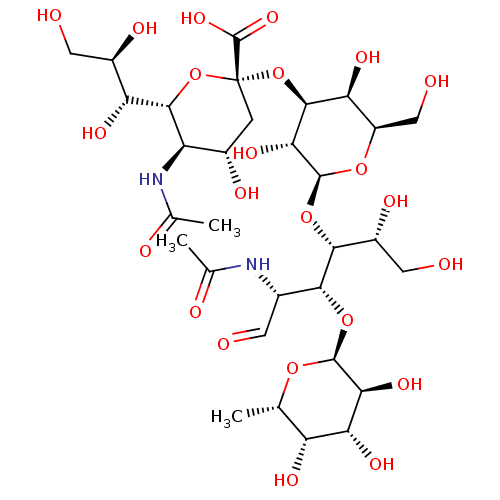
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Wada, Y; Saito, T; Matsuda, N; Ohmoto, H; Yoshino, K; Ohashi, M; Kondo, H; Ishida, H; Kiso, M; Hasegawa, A Studies on selectin blockers. 2. Novel selectin blocker as potential therapeutics for inflammatory disorders. J Med Chem39:2055-9 (1996) [PubMed] Article
Wada, Y; Saito, T; Matsuda, N; Ohmoto, H; Yoshino, K; Ohashi, M; Kondo, H; Ishida, H; Kiso, M; Hasegawa, A Studies on selectin blockers. 2. Novel selectin blocker as potential therapeutics for inflammatory disorders. J Med Chem39:2055-9 (1996) [PubMed] Article