

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
| Reaction Details | |||
|---|---|---|---|
 | Report a problem with these data | ||
| Target | Kappa-type opioid receptor | ||
| Ligand | BDBM50369225 | ||
| Substrate/Competitor | n/a | ||
| Meas. Tech. | ChEMBL_145552 (CHEMBL751016) | ||
| IC50 | 1.2±n/a nM | ||
| Citation |  Chang, AC; Chao, CC; Takemori, AE; Gekker, G; Hu, S; Peterson, PK; Portoghese, PS Arylacetamide-derived fluorescent probes: synthesis, biological evaluation, and direct fluorescent labeling of kappa opioid receptors in mouse microglial cells. J Med Chem39:1729-35 (1996) [PubMed] Article Chang, AC; Chao, CC; Takemori, AE; Gekker, G; Hu, S; Peterson, PK; Portoghese, PS Arylacetamide-derived fluorescent probes: synthesis, biological evaluation, and direct fluorescent labeling of kappa opioid receptors in mouse microglial cells. J Med Chem39:1729-35 (1996) [PubMed] Article | ||
| More Info.: | Get all data from this article, Assay Method | ||
| Kappa-type opioid receptor | |||
| Name: | Kappa-type opioid receptor | ||
| Synonyms: | KOR-1 | MSL-1 | OPIATE Kappa | OPRK_MOUSE | Oprk1 | ||
| Type: | Enzyme | ||
| Mol. Mass.: | 42656.78 | ||
| Organism: | Mus musculus (Mouse) | ||
| Description: | P33534 | ||
| Residue: | 380 | ||
| Sequence: |
| ||
| BDBM50369225 | |||
| n/a | |||
| Name | BDBM50369225 | ||
| Synonyms: | CHEMBL1907786 | ||
| Type | Small organic molecule | ||
| Emp. Form. | C42H36Cl2N4O6S | ||
| Mol. Mass. | 795.729 | ||
| SMILES | CN([C@H](CN1CCCC1)c1cccc(NC(=S)Nc2ccc(c(c2)C(O)=O)-c2c3ccc(O)cc3oc3cc(=O)ccc23)c1)C(=O)Cc1ccc(Cl)c(Cl)c1 |r,wU:2.1,(9.12,-13.32,;8.28,-12.01,;9.03,-10.64,;10.57,-10.59,;11.38,-11.87,;10.96,-13.34,;12.2,-14.2,;13.44,-13.29,;12.92,-11.83,;8.19,-9.33,;6.65,-9.4,;5.83,-8.11,;6.54,-6.75,;8.07,-6.68,;8.77,-5.29,;10.31,-5.21,;11.15,-6.51,;11.01,-3.83,;12.55,-3.76,;13.39,-5.04,;14.93,-4.95,;15.63,-3.57,;14.8,-2.32,;13.26,-2.39,;15.51,-.91,;17.06,-.82,;14.67,.37,;16.79,-4.62,;16.79,-6.16,;15.44,-6.93,;15.44,-8.47,;16.79,-9.24,;16.79,-10.78,;18.1,-8.47,;18.1,-6.93,;19.45,-6.16,;19.45,-4.62,;20.78,-3.85,;20.78,-2.32,;22.15,-1.52,;19.45,-1.55,;18.1,-2.32,;18.1,-3.85,;8.91,-7.96,;6.75,-12.06,;5.95,-10.78,;6.05,-13.44,;4.51,-13.5,;3.67,-12.2,;2.13,-12.26,;1.41,-13.6,;-.13,-13.64,;2.24,-14.92,;1.52,-16.3,;3.78,-14.86,)| | ||
| Structure | 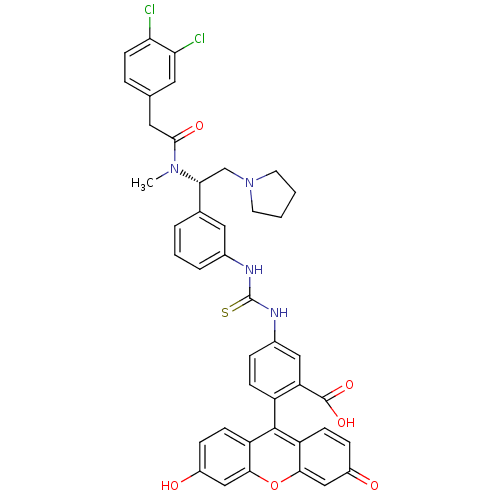
| ||