

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
| Reaction Details | |||
|---|---|---|---|
 | Report a problem with these data | ||
| Target | Prothrombin | ||
| Ligand | BDBM50060704 | ||
| Substrate/Competitor | n/a | ||
| Meas. Tech. | ChEMBL_208549 (CHEMBL814318) | ||
| Ki | 3±n/a nM | ||
| KON | 1800 M-1s-1 | ||
| Citation |  Tucker, TJ; Lumma, WC; Lewis, SD; Gardell, SJ; Lucas, BJ; Sisko, JT; Lynch, JJ; Lyle, EA; Baskin, EP; Woltmann, RF; Appleby, SD; Chen, IW; Dancheck, KB; Naylor-Olsen, AM; Krueger, JA; Cooper, CM; Vacca, JP Synthesis of a series of potent and orally bioavailable thrombin inhibitors that utilize 3,3-disubstituted propionic acid derivatives in the P3 position. J Med Chem40:3687-93 (1997) [PubMed] Article Tucker, TJ; Lumma, WC; Lewis, SD; Gardell, SJ; Lucas, BJ; Sisko, JT; Lynch, JJ; Lyle, EA; Baskin, EP; Woltmann, RF; Appleby, SD; Chen, IW; Dancheck, KB; Naylor-Olsen, AM; Krueger, JA; Cooper, CM; Vacca, JP Synthesis of a series of potent and orally bioavailable thrombin inhibitors that utilize 3,3-disubstituted propionic acid derivatives in the P3 position. J Med Chem40:3687-93 (1997) [PubMed] Article | ||
| More Info.: | Get all data from this article, Assay Method | ||
| Prothrombin | |||
| Name: | Prothrombin | ||
| Synonyms: | Activation peptide fragment 1 | Activation peptide fragment 2 | Coagulation factor II | F2 | Prothrombin precursor | THRB_HUMAN | Thrombin heavy chain | Thrombin light chain | ||
| Type: | Protein | ||
| Mol. Mass.: | 70029.57 | ||
| Organism: | Homo sapiens (Human) | ||
| Description: | P00734 | ||
| Residue: | 622 | ||
| Sequence: |
| ||
| BDBM50060704 | |||
| n/a | |||
| Name | BDBM50060704 | ||
| Synonyms: | (S)-1-(2-5H-Dibenzo[a,d]cyclohepten-5-yl-acetyl)-pyrrolidine-2-carboxylic acid (4-amino-cyclohexylmethyl)-amide; hydrochloride | CHEMBL555753 | ||
| Type | Small organic molecule | ||
| Emp. Form. | C29H35N3O2 | ||
| Mol. Mass. | 457.6071 | ||
| SMILES | NC1CCC(CNC(=O)[C@@H]2CCCN2C(=O)CC2c3ccccc3C=Cc3ccccc23)CC1 |wU:9.8,c:26,(10.27,-10.54,;9.32,-9.36,;8.93,-8.11,;9.57,-6.52,;8.23,-5.75,;6.86,-5.12,;6.7,-3.58,;5.27,-2.98,;4.05,-3.87,;5.12,-1.45,;6.29,-.43,;5.65,.98,;4.15,.82,;3.83,-.68,;2.42,-1.29,;2.27,-2.82,;1.18,-.39,;1.18,1.14,;-.35,1.17,;-.93,-.27,;-2.46,-.49,;-3.41,.72,;-2.84,2.16,;-1.31,2.38,;-.96,3.88,;.45,4.52,;1.82,3.82,;2.94,4.87,;4.4,4.42,;4.75,2.92,;3.61,1.87,;2.14,2.32,;8.65,-6.9,;8.01,-8.59,)| | ||
| Structure | 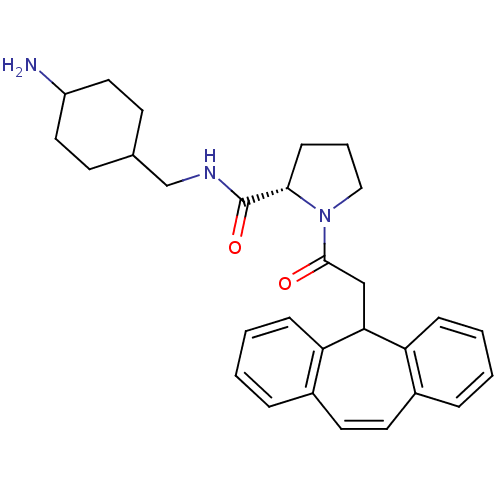
| ||