| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Adenosylhomocysteinase |
|---|
| Ligand | BDBM50369379 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_196552 (CHEMBL801947) |
|---|
| Ki | 3900±n/a nM |
|---|
| Citation |  Wnuk, SF; Mao, Y; Yuan, CS; Borchardt, RT; Andrei, G; Balzarini, J; De Clercq, E; Robins, MJ Discovery of type II (covalent) inactivation of S-adenosyl-L-homocysteine hydrolase involving its"hydrolytic activity": synthesis and evaluation of dihalohomovinyl nucleoside analogues derived from adenosine. J Med Chem41:3078-83 (1998) [PubMed] Article Wnuk, SF; Mao, Y; Yuan, CS; Borchardt, RT; Andrei, G; Balzarini, J; De Clercq, E; Robins, MJ Discovery of type II (covalent) inactivation of S-adenosyl-L-homocysteine hydrolase involving its"hydrolytic activity": synthesis and evaluation of dihalohomovinyl nucleoside analogues derived from adenosine. J Med Chem41:3078-83 (1998) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Adenosylhomocysteinase |
|---|
| Name: | Adenosylhomocysteinase |
|---|
| Synonyms: | AHCY | Adenosylhomocysteinase | SAHH | SAHH_HUMAN |
|---|
| Type: | PROTEIN |
|---|
| Mol. Mass.: | 47714.06 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | ChEMBL_1507791 |
|---|
| Residue: | 432 |
|---|
| Sequence: | MSDKLPYKVADIGLAAWGRKALDIAENEMPGLMRMRERYSASKPLKGARIAGCLHMTVET
AVLIETLVTLGAEVQWSSCNIFSTQDHAAAAIAKAGIPVYAWKGETDEEYLWCIEQTLYF
KDGPLNMILDDGGDLTNLIHTKYPQLLPGIRGISEETTTGVHNLYKMMANGILKVPAINV
NDSVTKSKFDNLYGCRESLIDGIKRATDVMIAGKVAVVAGYGDVGKGCAQALRGFGARVI
ITEIDPINALQAAMEGYEVTTMDEACQEGNIFVTTTGCIDIILGRHFEQMKDDAIVCNIG
HFDVEIDVKWLNENAVEKVNIKPQVDRYRLKNGRRIILLAEGRLVNLGCAMGHPSFVMSN
SFTNQVMAQIELWTHPDKYPVGVHFLPKKLDEAVAEAHLGKLNVKLTKLTEKQAQYLGMS
CDGPFKPDHYRY
|
|
|
|---|
| BDBM50369379 |
|---|
| n/a |
|---|
| Name | BDBM50369379 |
|---|
| Synonyms: | CHEMBL607755 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C11H11BrFN5O3 |
|---|
| Mol. Mass. | 360.139 |
|---|
| SMILES | Nc1ncnc2n(cnc12)C1O[C@H](\C=C(\F)Br)[C@@H](O)[C@H]1O |r| |
|---|
| Structure | 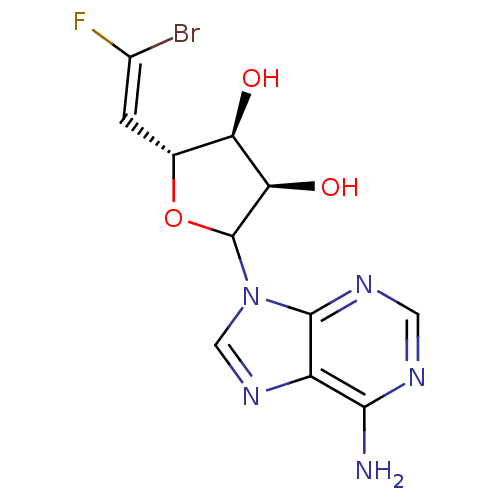
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Wnuk, SF; Mao, Y; Yuan, CS; Borchardt, RT; Andrei, G; Balzarini, J; De Clercq, E; Robins, MJ Discovery of type II (covalent) inactivation of S-adenosyl-L-homocysteine hydrolase involving its"hydrolytic activity": synthesis and evaluation of dihalohomovinyl nucleoside analogues derived from adenosine. J Med Chem41:3078-83 (1998) [PubMed] Article
Wnuk, SF; Mao, Y; Yuan, CS; Borchardt, RT; Andrei, G; Balzarini, J; De Clercq, E; Robins, MJ Discovery of type II (covalent) inactivation of S-adenosyl-L-homocysteine hydrolase involving its"hydrolytic activity": synthesis and evaluation of dihalohomovinyl nucleoside analogues derived from adenosine. J Med Chem41:3078-83 (1998) [PubMed] Article