| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Prothrombin |
|---|
| Ligand | BDBM50369433 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_208540 (CHEMBL813634) |
|---|
| Ki | 0.045000±n/a nM |
|---|
| Citation |  Quan, ML; Liauw, AY; Ellis, CD; Pruitt, JR; Carini, DJ; Bostrom, LL; Huang, PP; Harrison, K; Knabb, RM; Thoolen, MJ; Wong, PC; Wexler, RR Design and synthesis of isoxazoline derivatives as factor Xa inhibitors. 1. J Med Chem42:2752-9 (1999) [PubMed] Article Quan, ML; Liauw, AY; Ellis, CD; Pruitt, JR; Carini, DJ; Bostrom, LL; Huang, PP; Harrison, K; Knabb, RM; Thoolen, MJ; Wong, PC; Wexler, RR Design and synthesis of isoxazoline derivatives as factor Xa inhibitors. 1. J Med Chem42:2752-9 (1999) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Prothrombin |
|---|
| Name: | Prothrombin |
|---|
| Synonyms: | Activation peptide fragment 1 | Activation peptide fragment 2 | Coagulation factor II | F2 | Prothrombin precursor | THRB_HUMAN | Thrombin heavy chain | Thrombin light chain |
|---|
| Type: | Protein |
|---|
| Mol. Mass.: | 70029.57 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | P00734 |
|---|
| Residue: | 622 |
|---|
| Sequence: | MAHVRGLQLPGCLALAALCSLVHSQHVFLAPQQARSLLQRVRRANTFLEEVRKGNLEREC
VEETCSYEEAFEALESSTATDVFWAKYTACETARTPRDKLAACLEGNCAEGLGTNYRGHV
NITRSGIECQLWRSRYPHKPEINSTTHPGADLQENFCRNPDSSTTGPWCYTTDPTVRRQE
CSIPVCGQDQVTVAMTPRSEGSSVNLSPPLEQCVPDRGQQYQGRLAVTTHGLPCLAWASA
QAKALSKHQDFNSAVQLVENFCRNPDGDEEGVWCYVAGKPGDFGYCDLNYCEEAVEEETG
DGLDEDSDRAIEGRTATSEYQTFFNPRTFGSGEADCGLRPLFEKKSLEDKTERELLESYI
DGRIVEGSDAEIGMSPWQVMLFRKSPQELLCGASLISDRWVLTAAHCLLYPPWDKNFTEN
DLLVRIGKHSRTRYERNIEKISMLEKIYIHPRYNWRENLDRDIALMKLKKPVAFSDYIHP
VCLPDRETAASLLQAGYKGRVTGWGNLKETWTANVGKGQPSVLQVVNLPIVERPVCKDST
RIRITDNMFCAGYKPDEGKRGDACEGDSGGPFVMKSPFNNRWYQMGIVSWGEGCDRDGKY
GFYTHVFRLKKWIQKVIDQFGE
|
|
|
|---|
| BDBM50369433 |
|---|
| n/a |
|---|
| Name | BDBM50369433 |
|---|
| Synonyms: | CHEMBL1202108 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C21H33BN6O5 |
|---|
| Mol. Mass. | 460.335 |
|---|
| SMILES | [#6]-[#6](=O)-[#7]-[#6@H](-[#6]-c1ccccc1)-[#6](=O)-[#7]-1-[#6]-[#6]-[#6]-[#6@@H]-1-[#6](=O)-[#7]-[#6](-[#6]-[#6]-[#6]\[#7]=[#6](\[#7])-[#7])-[#5](-[#8])-[#8] |r| |
|---|
| Structure | 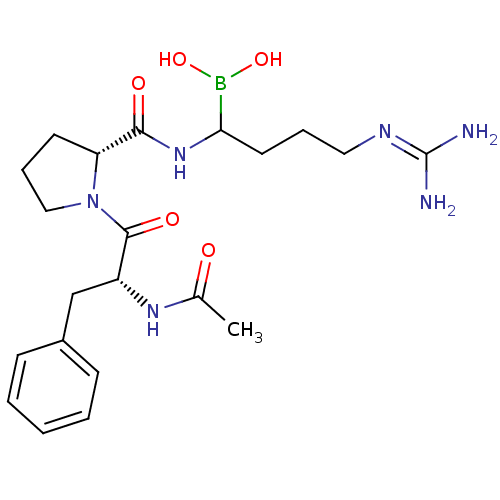
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Quan, ML; Liauw, AY; Ellis, CD; Pruitt, JR; Carini, DJ; Bostrom, LL; Huang, PP; Harrison, K; Knabb, RM; Thoolen, MJ; Wong, PC; Wexler, RR Design and synthesis of isoxazoline derivatives as factor Xa inhibitors. 1. J Med Chem42:2752-9 (1999) [PubMed] Article
Quan, ML; Liauw, AY; Ellis, CD; Pruitt, JR; Carini, DJ; Bostrom, LL; Huang, PP; Harrison, K; Knabb, RM; Thoolen, MJ; Wong, PC; Wexler, RR Design and synthesis of isoxazoline derivatives as factor Xa inhibitors. 1. J Med Chem42:2752-9 (1999) [PubMed] Article