| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Prothrombin |
|---|
| Ligand | BDBM50082611 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_210598 (CHEMBL816565) |
|---|
| Ki | 5.7±n/a nM |
|---|
| Citation |  Brundish, D; Bull, A; Donovan, V; Fullerton, JD; Garman, SM; Hayler, JF; Janus, D; Kane, PD; McDonnell, M; Smith, GP; Wakeford, R; Walker, CV; Howarth, G; Hoyle, W; Allen, MC; Ambler, J; Butler, K; Talbot, MD Design and synthesis of thrombin inhibitors: analogues of MD-805 with reduced stereogenicity and improved potency. J Med Chem42:4584-603 (1999) [PubMed] Brundish, D; Bull, A; Donovan, V; Fullerton, JD; Garman, SM; Hayler, JF; Janus, D; Kane, PD; McDonnell, M; Smith, GP; Wakeford, R; Walker, CV; Howarth, G; Hoyle, W; Allen, MC; Ambler, J; Butler, K; Talbot, MD Design and synthesis of thrombin inhibitors: analogues of MD-805 with reduced stereogenicity and improved potency. J Med Chem42:4584-603 (1999) [PubMed] |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Prothrombin |
|---|
| Name: | Prothrombin |
|---|
| Synonyms: | Activation peptide fragment 1 | Activation peptide fragment 2 | Coagulation factor II | F2 | Prothrombin | THRB_BOVIN | Thrombin | Thrombin heavy chain | Thrombin light chain |
|---|
| Type: | Enzyme |
|---|
| Mol. Mass.: | 70502.73 |
|---|
| Organism: | Bos taurus (Bovine) |
|---|
| Description: | P00735 |
|---|
| Residue: | 625 |
|---|
| Sequence: | MARVRGPRLPGCLALAALFSLVHSQHVFLAHQQASSLLQRARRANKGFLEEVRKGNLERE
CLEEPCSREEAFEALESLSATDAFWAKYTACESARNPREKLNECLEGNCAEGVGMNYRGN
VSVTRSGIECQLWRSRYPHKPEINSTTHPGADLRENFCRNPDGSITGPWCYTTSPTLRRE
ECSVPVCGQDRVTVEVIPRSGGSTTSQSPLLETCVPDRGREYRGRLAVTTSGSRCLAWSS
EQAKALSKDQDFNPAVPLAENFCRNPDGDEEGAWCYVADQPGDFEYCDLNYCEEPVDGDL
GDRLGEDPDPDAAIEGRTSEDHFQPFFNEKTFGAGEADCGLRPLFEKKQVQDQTEKELFE
SYIEGRIVEGQDAEVGLSPWQVMLFRKSPQELLCGASLISDRWVLTAAHCLLYPPWDKNF
TVDDLLVRIGKHSRTRYERKVEKISMLDKIYIHPRYNWKENLDRDIALLKLKRPIELSDY
IHPVCLPDKQTAAKLLHAGFKGRVTGWGNRRETWTTSVAEVQPSVLQVVNLPLVERPVCK
ASTRIRITDNMFCAGYKPGEGKRGDACEGDSGGPFVMKSPYNNRWYQMGIVSWGEGCDRD
GKYGFYTHVFRLKKWIQKVIDRLGS
|
|
|
|---|
| BDBM50082611 |
|---|
| n/a |
|---|
| Name | BDBM50082611 |
|---|
| Synonyms: | 3,3-Dimethyl-1,2,3,4-tetrahydro-quinoline-8-sulfonic acid {1-[4-(2-fluoro-ethyl)-piperidine-1-carbonyl]-4-guanidino-butyl}-amide | CHEMBL41519 | MD805 Analogue |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C24H39FN6O3S |
|---|
| Mol. Mass. | 510.668 |
|---|
| SMILES | CC1(C)CNc2c(C1)cccc2S(=O)(=O)NC(CCCNC(N)=N)C(=O)N1CCC(CCF)CC1 |
|---|
| Structure | 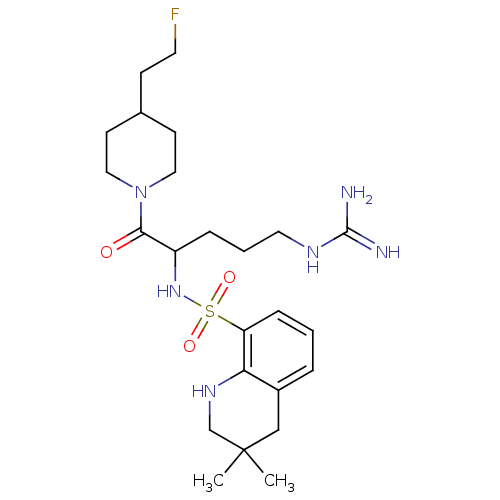
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Brundish, D; Bull, A; Donovan, V; Fullerton, JD; Garman, SM; Hayler, JF; Janus, D; Kane, PD; McDonnell, M; Smith, GP; Wakeford, R; Walker, CV; Howarth, G; Hoyle, W; Allen, MC; Ambler, J; Butler, K; Talbot, MD Design and synthesis of thrombin inhibitors: analogues of MD-805 with reduced stereogenicity and improved potency. J Med Chem42:4584-603 (1999) [PubMed]
Brundish, D; Bull, A; Donovan, V; Fullerton, JD; Garman, SM; Hayler, JF; Janus, D; Kane, PD; McDonnell, M; Smith, GP; Wakeford, R; Walker, CV; Howarth, G; Hoyle, W; Allen, MC; Ambler, J; Butler, K; Talbot, MD Design and synthesis of thrombin inhibitors: analogues of MD-805 with reduced stereogenicity and improved potency. J Med Chem42:4584-603 (1999) [PubMed]