| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Adenosine receptor A1 |
|---|
| Ligand | BDBM50207816 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_29113 (CHEMBL638725) |
|---|
| Ki | 1.3±n/a nM |
|---|
| Citation |  Bondavalli, F; Botta, M; Bruno, O; Ciacci, A; Corelli, F; Fossa, P; Lucacchini, A; Manetti, F; Martini, C; Menozzi, G; Mosti, L; Ranise, A; Schenone, S; Tafi, A; Trincavellic, ML Synthesis, molecular modeling studies, and pharmacological activity of selective A(1) receptor antagonists. J Med Chem45:4875-87 (2002) [PubMed] Bondavalli, F; Botta, M; Bruno, O; Ciacci, A; Corelli, F; Fossa, P; Lucacchini, A; Manetti, F; Martini, C; Menozzi, G; Mosti, L; Ranise, A; Schenone, S; Tafi, A; Trincavellic, ML Synthesis, molecular modeling studies, and pharmacological activity of selective A(1) receptor antagonists. J Med Chem45:4875-87 (2002) [PubMed] |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Adenosine receptor A1 |
|---|
| Name: | Adenosine receptor A1 |
|---|
| Synonyms: | AA1R_BOVIN | ADENOSINE A1 | ADENOSINE A1 high | ADENOSINE A1 low | ADORA1 | Adenosine A1 receptor |
|---|
| Type: | Enzyme Catalytic Domain |
|---|
| Mol. Mass.: | 36602.99 |
|---|
| Organism: | BOVINE |
|---|
| Description: | ADENOSINE 0 BOVINE::P28190 |
|---|
| Residue: | 326 |
|---|
| Sequence: | MPPSISAFQAAYIGIEVLIALVSVPGNVLVIWAVKVNQALRDATFCFIVSLAVADVAVGA
LVIPLAILINIGPRTYFHTCLKVACPVLILTQSSILALLAIAVDRYLRVKIPLRYKTVVT
PRRAVVAITGCWILSFVVGLTPMFGWNNLSAVERDWLANGSVGEPVIECQFEKVISMEYM
VYFNFFVWVLPPLLLMVLIYMEVFYLIRKQLNKKVSASSGDPQKYYGKELKIAKSLALIL
FLFALSWLPLHILNCITLFCPSCHMPRILIYIAIFLSHGNSAMNPIVYAFRIQKFRVTFL
KIWNDHFRCQPAPPVDEDAPAERPDD
|
|
|
|---|
| BDBM50207816 |
|---|
| n/a |
|---|
| Name | BDBM50207816 |
|---|
| Synonyms: | CHEMBL273094 | N-(2-Amino-ethyl)-2-[4-(2,6-dioxo-1,3-dipropyl-2,3,6,7-tetrahydro-1H-purin-8-yl)-phenoxy]-acetamide | N-(2-Amino-ethyl)-2-[4-(2,6-dioxo-1,3-dipropyl-2,3,6,7-tetrahydro-1H-purin-8-yl)-phenoxy]-acetamide (xanthine amine congenere, XAC) | N-(2-Amino-ethyl)-2-[4-(2,6-dioxo-1,3-dipropyl-2,3,6,7-tetrahydro-1H-purin-8-yl)-phenoxy]-acetamide(XAC) | N-(2-Amino-ethyl)-2-[4-(2,6-dioxo-1,3-dipropyl-2,3,6,9-tetrahydro-1H-purin-8-yl)-phenoxy]-acetamide | N-(2-aminoethyl)-2-(4-(2,6-dioxo-1,3-dipropyl-2,3,6,7-tetrahydro-1H-purin-8-yl)phenoxy)acetamide | XAC |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C21H28N6O4 |
|---|
| Mol. Mass. | 428.4848 |
|---|
| SMILES | CCCn1c2nc([nH]c2c(=O)n(CCC)c1=O)-c1ccc(OCC(=O)NCCN)cc1 |
|---|
| Structure | 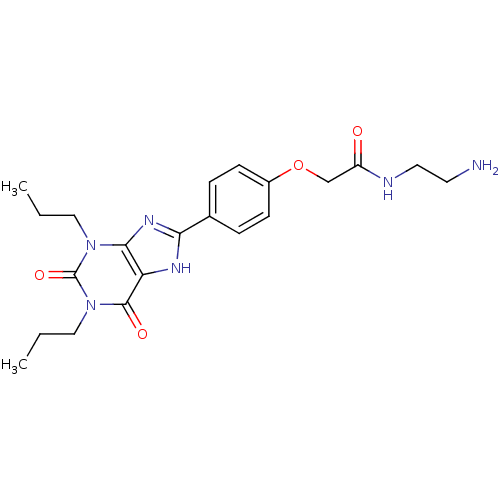
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Bondavalli, F; Botta, M; Bruno, O; Ciacci, A; Corelli, F; Fossa, P; Lucacchini, A; Manetti, F; Martini, C; Menozzi, G; Mosti, L; Ranise, A; Schenone, S; Tafi, A; Trincavellic, ML Synthesis, molecular modeling studies, and pharmacological activity of selective A(1) receptor antagonists. J Med Chem45:4875-87 (2002) [PubMed]
Bondavalli, F; Botta, M; Bruno, O; Ciacci, A; Corelli, F; Fossa, P; Lucacchini, A; Manetti, F; Martini, C; Menozzi, G; Mosti, L; Ranise, A; Schenone, S; Tafi, A; Trincavellic, ML Synthesis, molecular modeling studies, and pharmacological activity of selective A(1) receptor antagonists. J Med Chem45:4875-87 (2002) [PubMed]