| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Epidermal growth factor receptor |
|---|
| Ligand | BDBM50122567 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_66597 (CHEMBL680028) |
|---|
| IC50 | 310±n/a nM |
|---|
| Citation |  Wissner, A; Overbeek, E; Reich, MF; Floyd, MB; Johnson, BD; Mamuya, N; Rosfjord, EC; Discafani, C; Davis, R; Shi, X; Rabindran, SK; Gruber, BC; Ye, F; Hallett, WA; Nilakantan, R; Shen, R; Wang, YF; Greenberger, LM; Tsou, HR Synthesis and structure-activity relationships of 6,7-disubstituted 4-anilinoquinoline-3-carbonitriles. The design of an orally active, irreversible inhibitor of the tyrosine kinase activity of the epidermal growth factor receptor (EGFR) and the human epidermal growth factor receptor-2 (HER-2). J Med Chem46:49-63 (2002) [PubMed] Article Wissner, A; Overbeek, E; Reich, MF; Floyd, MB; Johnson, BD; Mamuya, N; Rosfjord, EC; Discafani, C; Davis, R; Shi, X; Rabindran, SK; Gruber, BC; Ye, F; Hallett, WA; Nilakantan, R; Shen, R; Wang, YF; Greenberger, LM; Tsou, HR Synthesis and structure-activity relationships of 6,7-disubstituted 4-anilinoquinoline-3-carbonitriles. The design of an orally active, irreversible inhibitor of the tyrosine kinase activity of the epidermal growth factor receptor (EGFR) and the human epidermal growth factor receptor-2 (HER-2). J Med Chem46:49-63 (2002) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Epidermal growth factor receptor |
|---|
| Name: | Epidermal growth factor receptor |
|---|
| Synonyms: | EGF Receptor Substrate | EGFR | EGFR_HUMAN | ERBB | ERBB1 | Epidermal Growth Factor Receptor Tyrosine Kinase | Epidermal Growth Factor Receptor erbB-1 | Epidermal Growth Factor Receptor, ErbB-1 | Epidermal growth factor receptor (EGFR) | Epidermal growth factor receptor (HER1) | HER1 | Protein-Tyrosine Kinase Erbb-1 | Proto-oncogene c-ErbB-1 | Receptor tyrosine-protein kinase ErbB-1 | Tyrosine-Protein Kinase ErbB-1 |
|---|
| Type: | Receptor Kinase Domain |
|---|
| Mol. Mass.: | 134279.59 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | P00533 |
|---|
| Residue: | 1210 |
|---|
| Sequence: | MRPSGTAGAALLALLAALCPASRALEEKKVCQGTSNKLTQLGTFEDHFLSLQRMFNNCEV
VLGNLEITYVQRNYDLSFLKTIQEVAGYVLIALNTVERIPLENLQIIRGNMYYENSYALA
VLSNYDANKTGLKELPMRNLQEILHGAVRFSNNPALCNVESIQWRDIVSSDFLSNMSMDF
QNHLGSCQKCDPSCPNGSCWGAGEENCQKLTKIICAQQCSGRCRGKSPSDCCHNQCAAGC
TGPRESDCLVCRKFRDEATCKDTCPPLMLYNPTTYQMDVNPEGKYSFGATCVKKCPRNYV
VTDHGSCVRACGADSYEMEEDGVRKCKKCEGPCRKVCNGIGIGEFKDSLSINATNIKHFK
NCTSISGDLHILPVAFRGDSFTHTPPLDPQELDILKTVKEITGFLLIQAWPENRTDLHAF
ENLEIIRGRTKQHGQFSLAVVSLNITSLGLRSLKEISDGDVIISGNKNLCYANTINWKKL
FGTSGQKTKIISNRGENSCKATGQVCHALCSPEGCWGPEPRDCVSCRNVSRGRECVDKCN
LLEGEPREFVENSECIQCHPECLPQAMNITCTGRGPDNCIQCAHYIDGPHCVKTCPAGVM
GENNTLVWKYADAGHVCHLCHPNCTYGCTGPGLEGCPTNGPKIPSIATGMVGALLLLLVV
ALGIGLFMRRRHIVRKRTLRRLLQERELVEPLTPSGEAPNQALLRILKETEFKKIKVLGS
GAFGTVYKGLWIPEGEKVKIPVAIKELREATSPKANKEILDEAYVMASVDNPHVCRLLGI
CLTSTVQLITQLMPFGCLLDYVREHKDNIGSQYLLNWCVQIAKGMNYLEDRRLVHRDLAA
RNVLVKTPQHVKITDFGLAKLLGAEEKEYHAEGGKVPIKWMALESILHRIYTHQSDVWSY
GVTVWELMTFGSKPYDGIPASEISSILEKGERLPQPPICTIDVYMIMVKCWMIDADSRPK
FRELIIEFSKMARDPQRYLVIQGDERMHLPSPTDSNFYRALMDEEDMDDVVDADEYLIPQ
QGFFSSPSTSRTPLLSSLSATSNNSTVACIDRNGLQSCPIKEDSFLQRYSSDPTGALTED
SIDDTFLPVPEYINQSVPKRPAGSVQNPVYHNQPLNPAPSRDPHYQDPHSTAVGNPEYLN
TVQPTCVNSTFDSPAHWAQKGSHQISLDNPDYQQDFFPKEAKPNGIFKGSTAENAEYLRV
APQSSEFIGA
|
|
|
|---|
| BDBM50122567 |
|---|
| n/a |
|---|
| Name | BDBM50122567 |
|---|
| Synonyms: | (E)-4-Dimethylamino-but-2-enoic acid [4-(3-bromo-phenylamino)-3-cyano-quinolin-6-yl]-amide | 4-Dimethylamino-but-2-enoic acid [4-(3-bromo-phenylamino)-3-cyano-quinolin-6-yl]-amide | CHEMBL30973 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C22H20BrN5O |
|---|
| Mol. Mass. | 450.331 |
|---|
| SMILES | CN(C)C\C=C\C(=O)Nc1ccc2ncc(C#N)c(Nc3cccc(Br)c3)c2c1 |
|---|
| Structure | 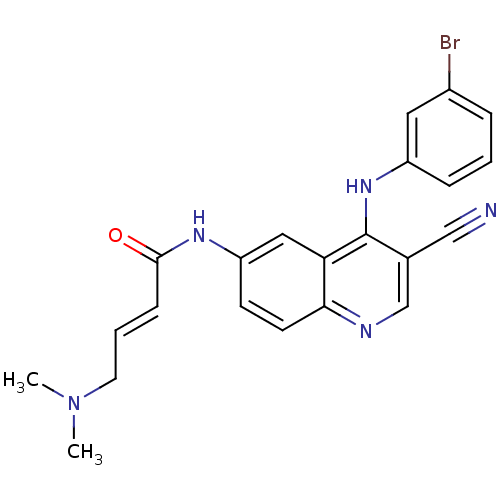
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Wissner, A; Overbeek, E; Reich, MF; Floyd, MB; Johnson, BD; Mamuya, N; Rosfjord, EC; Discafani, C; Davis, R; Shi, X; Rabindran, SK; Gruber, BC; Ye, F; Hallett, WA; Nilakantan, R; Shen, R; Wang, YF; Greenberger, LM; Tsou, HR Synthesis and structure-activity relationships of 6,7-disubstituted 4-anilinoquinoline-3-carbonitriles. The design of an orally active, irreversible inhibitor of the tyrosine kinase activity of the epidermal growth factor receptor (EGFR) and the human epidermal growth factor receptor-2 (HER-2). J Med Chem46:49-63 (2002) [PubMed] Article
Wissner, A; Overbeek, E; Reich, MF; Floyd, MB; Johnson, BD; Mamuya, N; Rosfjord, EC; Discafani, C; Davis, R; Shi, X; Rabindran, SK; Gruber, BC; Ye, F; Hallett, WA; Nilakantan, R; Shen, R; Wang, YF; Greenberger, LM; Tsou, HR Synthesis and structure-activity relationships of 6,7-disubstituted 4-anilinoquinoline-3-carbonitriles. The design of an orally active, irreversible inhibitor of the tyrosine kinase activity of the epidermal growth factor receptor (EGFR) and the human epidermal growth factor receptor-2 (HER-2). J Med Chem46:49-63 (2002) [PubMed] Article