| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Cannabinoid receptor 1 |
|---|
| Ligand | BDBM50272598 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_46450 (CHEMBL657898) |
|---|
| Ki | 16±n/a nM |
|---|
| Citation |  Wrobleski, ST; Chen, P; Hynes, J; Lin, S; Norris, DJ; Pandit, CR; Spergel, S; Wu, H; Tokarski, JS; Chen, X; Gillooly, KM; Kiener, PA; McIntyre, KW; Patil-Koota, V; Shuster, DJ; Turk, LA; Yang, G; Leftheris, K Rational design and synthesis of an orally active indolopyridone as a novel conformationally constrained cannabinoid ligand possessing antiinflammatory properties. J Med Chem46:2110-6 (2003) [PubMed] Article Wrobleski, ST; Chen, P; Hynes, J; Lin, S; Norris, DJ; Pandit, CR; Spergel, S; Wu, H; Tokarski, JS; Chen, X; Gillooly, KM; Kiener, PA; McIntyre, KW; Patil-Koota, V; Shuster, DJ; Turk, LA; Yang, G; Leftheris, K Rational design and synthesis of an orally active indolopyridone as a novel conformationally constrained cannabinoid ligand possessing antiinflammatory properties. J Med Chem46:2110-6 (2003) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Cannabinoid receptor 1 |
|---|
| Name: | Cannabinoid receptor 1 |
|---|
| Synonyms: | CANN6 | CANNABINOID CB1 | CB-R | CB1 | CNR | CNR1 | CNR1_HUMAN | Cannabinoid CB1 receptor | Cannabinoid receptor | Cannabinoid receptor 1 (CB1) | Cannabinoid receptor 1 (brain) |
|---|
| Type: | G Protein-Coupled Receptor (GPCR) |
|---|
| Mol. Mass.: | 52868.96 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | P21554 |
|---|
| Residue: | 472 |
|---|
| Sequence: | MKSILDGLADTTFRTITTDLLYVGSNDIQYEDIKGDMASKLGYFPQKFPLTSFRGSPFQE
KMTAGDNPQLVPADQVNITEFYNKSLSSFKENEENIQCGENFMDIECFMVLNPSQQLAIA
VLSLTLGTFTVLENLLVLCVILHSRSLRCRPSYHFIGSLAVADLLGSVIFVYSFIDFHVF
HRKDSRNVFLFKLGGVTASFTASVGSLFLTAIDRYISIHRPLAYKRIVTRPKAVVAFCLM
WTIAIVIAVLPLLGWNCEKLQSVCSDIFPHIDETYLMFWIGVTSVLLLFIVYAYMYILWK
AHSHAVRMIQRGTQKSIIIHTSEDGKVQVTRPDQARMDIRLAKTLVLILVVLIICWGPLL
AIMVYDVFGKMNKLIKTVFAFCSMLCLLNSTVNPIIYALRSKDLRHAFRSMFPSCEGTAQ
PLDNSMGDSDCLHKHANNAASVHRAAESCIKSTVKIAKVTMSVSTDTSAEAL
|
|
|
|---|
| BDBM50272598 |
|---|
| n/a |
|---|
| Name | BDBM50272598 |
|---|
| Synonyms: | 6-Methoxy-5-(2-morpholin-4-yl-ethyl)-2-(1,3,3-trimethyl-bicyclo[2.2.1]hept-2-yl)-2,5-dihydro-pyrido[4,3-b]indol-1-one | 6-methoxy-5-(2-morpholinoethyl)-2-((1S,2S,4R)-1,3,3-trimethylbicyclo[2.2.1]heptan-2-yl)-2H-pyrido[4,3-b]indol-1(5H)-one | CHEMBL527031 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C28H37N3O3 |
|---|
| Mol. Mass. | 463.6117 |
|---|
| SMILES | COc1cccc2c1n(CCN1CCOCC1)c1ccn([C@H]3[C@@]4(C)CC[C@H](C4)C3(C)C)c(=O)c21 |r| |
|---|
| Structure | 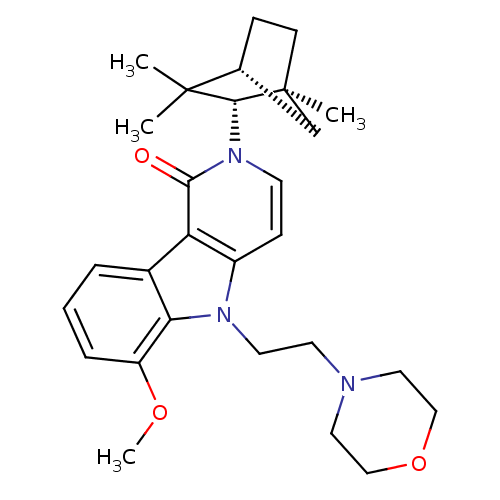
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Wrobleski, ST; Chen, P; Hynes, J; Lin, S; Norris, DJ; Pandit, CR; Spergel, S; Wu, H; Tokarski, JS; Chen, X; Gillooly, KM; Kiener, PA; McIntyre, KW; Patil-Koota, V; Shuster, DJ; Turk, LA; Yang, G; Leftheris, K Rational design and synthesis of an orally active indolopyridone as a novel conformationally constrained cannabinoid ligand possessing antiinflammatory properties. J Med Chem46:2110-6 (2003) [PubMed] Article
Wrobleski, ST; Chen, P; Hynes, J; Lin, S; Norris, DJ; Pandit, CR; Spergel, S; Wu, H; Tokarski, JS; Chen, X; Gillooly, KM; Kiener, PA; McIntyre, KW; Patil-Koota, V; Shuster, DJ; Turk, LA; Yang, G; Leftheris, K Rational design and synthesis of an orally active indolopyridone as a novel conformationally constrained cannabinoid ligand possessing antiinflammatory properties. J Med Chem46:2110-6 (2003) [PubMed] Article