| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Alpha-2A adrenergic receptor [16-465] |
|---|
| Ligand | BDBM50131922 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_32765 (CHEMBL643710) |
|---|
| Ki | 76±n/a nM |
|---|
| Citation |  Campiani, G; Butini, S; Trotta, F; Fattorusso, C; Catalanotti, B; Aiello, F; Gemma, S; Nacci, V; Novellino, E; Stark, JA; Cagnotto, A; Fumagalli, E; Carnovali, F; Cervo, L; Mennini, T Synthesis and pharmacological evaluation of potent and highly selective D3 receptor ligands: inhibition of cocaine-seeking behavior and the role of dopamine D3/D2 receptors. J Med Chem46:3822-39 (2003) [PubMed] Article Campiani, G; Butini, S; Trotta, F; Fattorusso, C; Catalanotti, B; Aiello, F; Gemma, S; Nacci, V; Novellino, E; Stark, JA; Cagnotto, A; Fumagalli, E; Carnovali, F; Cervo, L; Mennini, T Synthesis and pharmacological evaluation of potent and highly selective D3 receptor ligands: inhibition of cocaine-seeking behavior and the role of dopamine D3/D2 receptors. J Med Chem46:3822-39 (2003) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Alpha-2A adrenergic receptor [16-465] |
|---|
| Name: | Alpha-2A adrenergic receptor [16-465] |
|---|
| Synonyms: | ADA2A_RAT | Adra2a | Adrenaline 2 | Alpha-2A adrenergic receptor | Alpha-2A adrenoceptor | Alpha-2A adrenoreceptor | Alpha-2AAR | Alpha-2D adrenergic receptor | Alpha2 Adrenoreceptor | CA2-47 | adrenergic Alpha2A |
|---|
| Type: | G-protein coupled receptor |
|---|
| Mol. Mass.: | 48961.69 |
|---|
| Organism: | Rattus norvegicus (rat) |
|---|
| Description: | P22909[16-465] |
|---|
| Residue: | 450 |
|---|
| Sequence: | MGSLQPDAGNSSWNGTEAPGGGTRATPYSLQVTLTLVCLAGLLMLFTVFGNVLVIIAVFT
SRALKAPQNLFLVSLASADILVATLVIPFSLANEVMGYWYFGKVWCEIYLALDVLFCTSS
IVHLCAISLDRYWSITQAIEYNLKRTPRRIKAIIVTVWVISAVISFPPLISIEKKGAGGG
QQPAEPSCKINDQKWYVISSSIGSFFAPCLIMILVYVRIYQIAKRRTRVPPSRRGPDACS
APPGGADRRPNGLGPERGAGTAGAEAEPLPTQLNGAPGEPAPTRPRDGDALDLEESSSSE
HAERPQGPGKPERGPRAKGKTKASQVKPGDSLPRRGPGAAGPGASGSGQGEERAGGAKAS
RWRGRQNREKRFTFVLAVVIGVFVVCWFPFFFTYTLIAVGCPVPYQLFNFFFWFGYCNSS
LNPVIYTIFNHDFRRAFKKILCRGDRKRIV
|
|
|
|---|
| BDBM50131922 |
|---|
| n/a |
|---|
| Name | BDBM50131922 |
|---|
| Synonyms: | 1H-Indole-2-carboxylic acid {4-[4-(2-methoxy-phenyl)-piperazin-1-yl]-butyl}-amide | CHEMBL125916 | N-(4-(4-(2-methoxyphenyl)piperazin-1-yl)butyl)-1H-indole-2-carboxamide |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C24H30N4O2 |
|---|
| Mol. Mass. | 406.5206 |
|---|
| SMILES | COc1ccccc1N1CCN(CCCCNC(=O)c2cc3ccccc3[nH]2)CC1 |
|---|
| Structure | 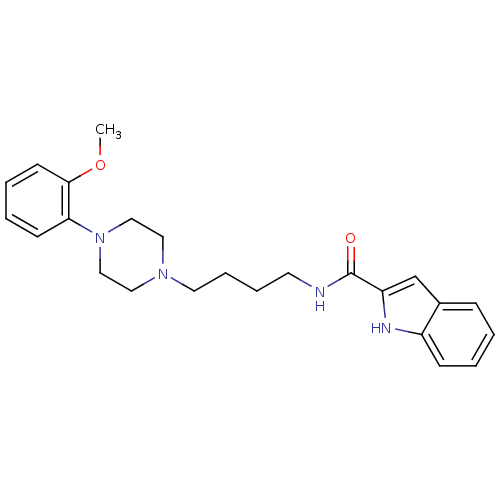
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Campiani, G; Butini, S; Trotta, F; Fattorusso, C; Catalanotti, B; Aiello, F; Gemma, S; Nacci, V; Novellino, E; Stark, JA; Cagnotto, A; Fumagalli, E; Carnovali, F; Cervo, L; Mennini, T Synthesis and pharmacological evaluation of potent and highly selective D3 receptor ligands: inhibition of cocaine-seeking behavior and the role of dopamine D3/D2 receptors. J Med Chem46:3822-39 (2003) [PubMed] Article
Campiani, G; Butini, S; Trotta, F; Fattorusso, C; Catalanotti, B; Aiello, F; Gemma, S; Nacci, V; Novellino, E; Stark, JA; Cagnotto, A; Fumagalli, E; Carnovali, F; Cervo, L; Mennini, T Synthesis and pharmacological evaluation of potent and highly selective D3 receptor ligands: inhibition of cocaine-seeking behavior and the role of dopamine D3/D2 receptors. J Med Chem46:3822-39 (2003) [PubMed] Article