| Reaction Details |
|---|
 | Report a problem with these data |
| Target | 5-hydroxytryptamine receptor 1A |
|---|
| Ligand | BDBM78940 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_894 (CHEMBL615805) |
|---|
| Ki | 2.2±n/a nM |
|---|
| Citation |  Balle, T; Perregaard, J; Ramirez, MT; Larsen, AK; Søby, KK; Liljefors, T; Andersen, K Synthesis and structure-affinity relationship investigations of 5-heteroaryl-substituted analogues of the antipsychotic sertindole. A new class of highly selective alpha(1) adrenoceptor antagonists. J Med Chem46:265-83 (2003) [PubMed] Article Balle, T; Perregaard, J; Ramirez, MT; Larsen, AK; Søby, KK; Liljefors, T; Andersen, K Synthesis and structure-affinity relationship investigations of 5-heteroaryl-substituted analogues of the antipsychotic sertindole. A new class of highly selective alpha(1) adrenoceptor antagonists. J Med Chem46:265-83 (2003) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| 5-hydroxytryptamine receptor 1A |
|---|
| Name: | 5-hydroxytryptamine receptor 1A |
|---|
| Synonyms: | 5-HT-1A | 5-HT1A | 5-hydroxytryptamine receptor 1A (5-HT-1A) | 5HT1A_HUMAN | ADRB2RL1 | ADRBRL1 | Dopamine D2 receptor and serotonin 1a receptor | G-21 | HTR1A | Serotonin receptor 1A |
|---|
| Type: | n/a |
|---|
| Mol. Mass.: | 46122.49 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | n/a |
|---|
| Residue: | 422 |
|---|
| Sequence: | MDVLSPGQGNNTTSPPAPFETGGNTTGISDVTVSYQVITSLLLGTLIFCAVLGNACVVAA
IALERSLQNVANYLIGSLAVTDLMVSVLVLPMAALYQVLNKWTLGQVTCDLFIALDVLCC
TSSILHLCAIALDRYWAITDPIDYVNKRTPRRAAALISLTWLIGFLISIPPMLGWRTPED
RSDPDACTISKDHGYTIYSTFGAFYIPLLLMLVLYGRIFRAARFRIRKTVKKVEKTGADT
RHGASPAPQPKKSVNGESGSRNWRLGVESKAGGALCANGAVRQGDDGAALEVIEVHRVGN
SKEHLPLPSEAGPTPCAPASFERKNERNAEAKRKMALARERKTVKTLGIIMGTFILCWLP
FFIVALVLPFCESSCHMPTLLGAIINWLGYSNSLLNPVIYAYFNKDFQNAFKKIIKCKFC
RQ
|
|
|
|---|
| BDBM78940 |
|---|
| n/a |
|---|
| Name | BDBM78940 |
|---|
| Synonyms: | METHIOTHEPIN | MLS000859918 | Methiothepin mesylate salt | SMR000326779 | cid_3039995 | mesylic acid;1-methyl-4-[3-(methylthio)-5,6-dihydrobenzo[b][1]benzothiepin-5-yl]piperazine | methanesulfonic acid;1-methyl-4-(3-methylsulfanyl-5,6-dihydrobenzo[b][1]benzothiepin-5-yl)piperazine | methanesulfonic acid;1-methyl-4-[3-(methylthio)-5,6-dihydrobenzo[b][1]benzothiepin-5-yl]piperazine |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C20H24N2S2 |
|---|
| Mol. Mass. | 356.548 |
|---|
| SMILES | CSc1ccc2Sc3ccccc3CC(N3CCN(C)CC3)c2c1 |
|---|
| Structure | 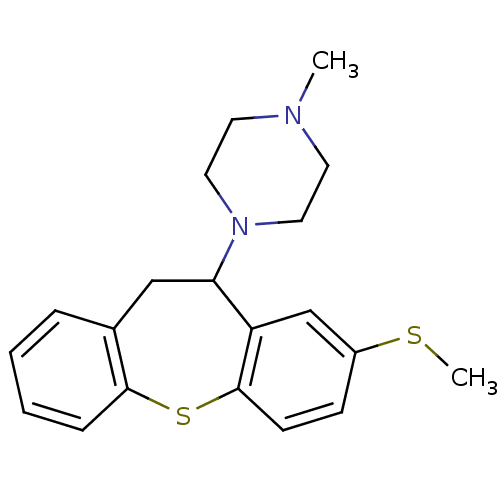
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Balle, T; Perregaard, J; Ramirez, MT; Larsen, AK; Søby, KK; Liljefors, T; Andersen, K Synthesis and structure-affinity relationship investigations of 5-heteroaryl-substituted analogues of the antipsychotic sertindole. A new class of highly selective alpha(1) adrenoceptor antagonists. J Med Chem46:265-83 (2003) [PubMed] Article
Balle, T; Perregaard, J; Ramirez, MT; Larsen, AK; Søby, KK; Liljefors, T; Andersen, K Synthesis and structure-affinity relationship investigations of 5-heteroaryl-substituted analogues of the antipsychotic sertindole. A new class of highly selective alpha(1) adrenoceptor antagonists. J Med Chem46:265-83 (2003) [PubMed] Article